What the Whooping Cough Numbers Actually Show
A plain-language review of two vaccine strategies for protecting newborns, with the costs included
The United States government recommends that pregnant women receive the Tdap vaccine (tetanus, diphtheria, and acellular pertussis) during every pregnancy. It also recommends that parents, grandparents, and caregivers of newborns get vaccinated, a strategy known as cocooning. The rationale is straightforward: whooping cough kills infants, infants cannot be vaccinated until two months of age, and these two strategies may provide a protective bridge during that gap.
What is less straightforward is the evidence behind these recommendations, and what it actually tells us about benefit, cost, and risk. A detailed comparative review article attached below this summary examines both strategies side by side using the metrics that clinicians and health economists use internally but that public health communication routinely omits: absolute risk reduction, number needed to vaccinate, cost per outcome prevented, and number needed to harm. The findings are more nuanced than the official messaging suggests.
This summary presents those findings in plain language, with objective, fact-based analysis of both the strategies being promoted and the institutional confidence with which they are marketed and endorsed by CDC and ACIP.
THE PROBLEM
Why Newborns Are Vulnerable, and Why the Numbers Matter
Whooping cough is most dangerous in the first three months of life. The standard childhood vaccination schedule does not begin until two months of age, leaving every newborn unprotected for their most vulnerable window. In the United States between 2013 and 2022, 57% of pertussis-related deaths occurred in infants younger than two months.
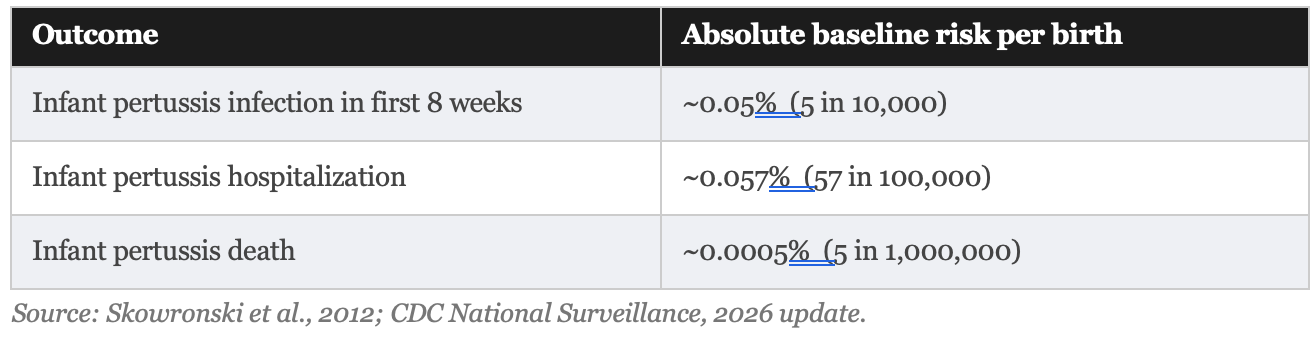
But before we assess any solution, we need to know how large the problem actually is in absolute terms: not in relative terms, not in emotional terms, but in raw numbers. The current risk figures in the contemporary United States are:
These are not large numbers. In a country of roughly 3.6 million births per year, approximately 1,800 infants contract pertussis in the first eight weeks, roughly 2,000 are hospitalized for it across the full first year, and somewhere between 5 and 18 infants die from it annually. That is devastating for the families involved, but rare in population terms. This context is not a reason to dismiss the strategies. It is a reason to evaluate them honestly.
STRATEGY ONE
Cocooning: The Idea That Never Really Worked
The cocooning strategy was endorsed by the Advisory Committee on Immunization Practices in 2005. The idea is intuitive: vaccinate everyone in close contact with the newborn, and you create a protective ring of immune people around the baby. In theory, it is compelling. In practice, it has largely failed.
Why It Fails in Theory
For cocooning to work, two things must be true. First, enough of the infant’s infections must originate from vaccinated contacts for the vaccination to make a difference. Studies estimate that household contacts account for 35% to 75% of infant infections, a wide and uncertain range. The remaining 25% to 65% come from casual contacts that cocooning cannot reach at all.
Second, the adult whooping cough booster (Tdap) must maintain sufficient protection long enough to matter. Here the evidence is discouraging. Studies show the booster provides roughly 69% protection in the first year, falling to less than 9% after four years. Parents vaccinated at the birth of their first child offer almost no protection by the time a second child arrives.
Why It Fails in Practice
To achieve meaningful herd protection through cocooning, vaccination coverage among all close contacts would need to reach 92% to 94%. No published program has come close to this. Real-world data from Switzerland found vaccination rates among parents ranged from 61% to 69%, dropping to 18% among uncles, 13% among aunts, and just 7% among grandparents. The coverage required for the theory to hold does not exist in the real world.
By 2011, the ACIP itself acknowledged that cocooning alone was insufficient to prevent pertussis morbidity and mortality in newborns. That conclusion, rendered more than a decade ago, has not prevented continued official endorsement of the strategy as a supplement.
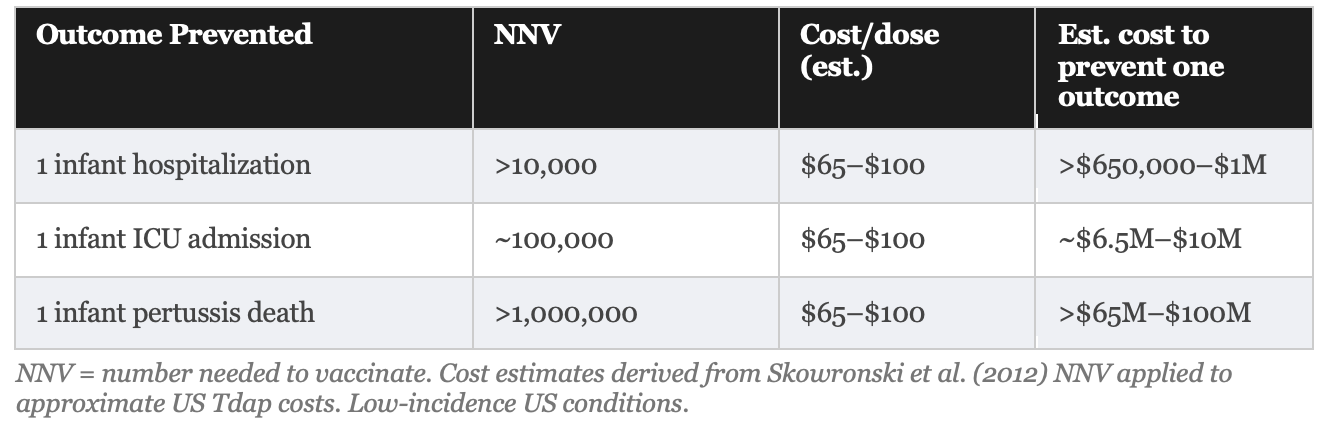
The Numbers: What Cocooning Would Cost to Prevent One Death
“To prevent one infant pertussis death through parental cocooning, more than one million parents would need to be vaccinated under low-incidence conditions.”
Skowronski et al., Clinical Infectious Diseases, 2012
This figure, derived from Canadian provincial surveillance data using a transparent formula, is not an outlier. It reflects the mathematical reality of multiplying a very low absolute baseline mortality rate (less than 0.0005% per birth) by a strategy that addresses only a fraction of sources. A parallel Italian analysis reached the same conclusion.
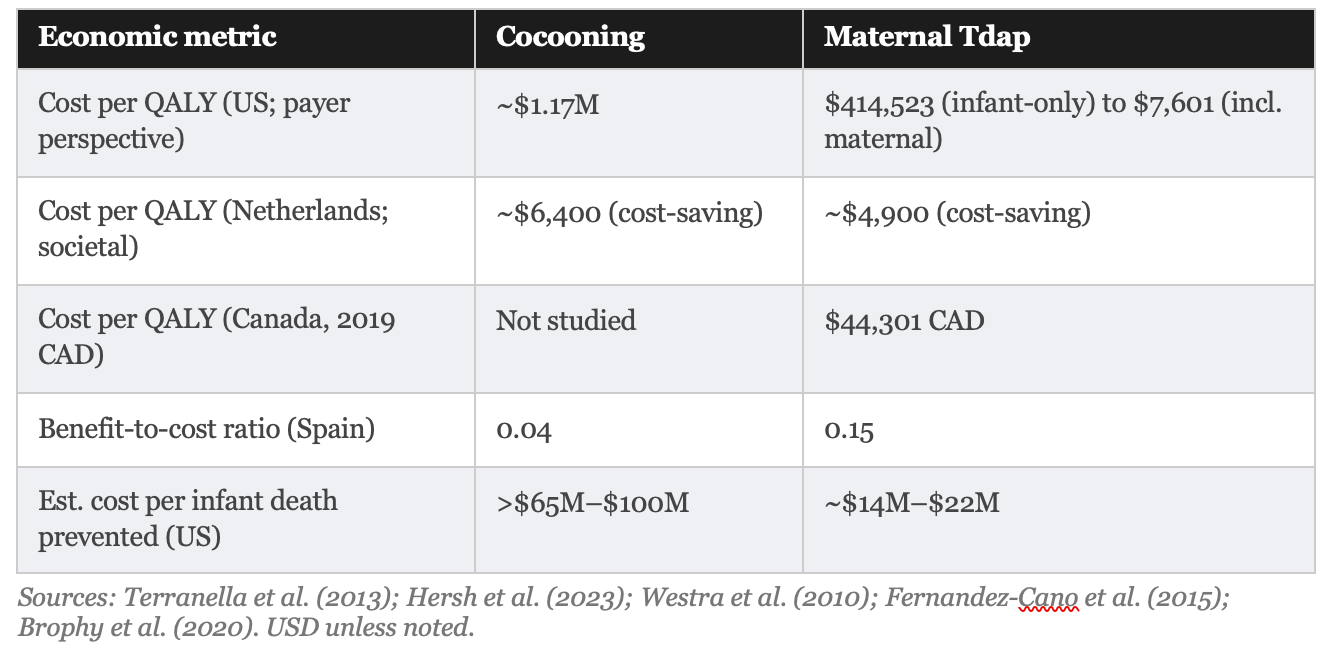
Published cost-effectiveness analyses confirm this picture. In the most detailed US comparison, Terranella and colleagues found cocooning costs approximately $1,172,825 per quality-adjusted life year (QALY) saved, which is roughly seven times the standard US willingness-to-pay threshold of $100,000 to $159,429 per QALY. A Spanish cost-benefit analysis found a return of just $0.04 for every $1.00 spent on cocooning.
A federal class action lawsuit illustrates precisely this gap between advertising claim and scientific reality. In DeCostanzo v. GlaxoSmithKline (E.D.N.Y., filed 2020), lead counsel Aaron Siri of Siri & Glimstad LLP brought a class action against GlaxoSmithKline (GSK) over the company’s “Big Bad Cough” advertising campaign for its Boostrix Tdap vaccine. The campaign ran from 2015 to 2020 and featured anthropomorphic wolves, including a grandparent who transforms into a wolf upon cradling a newborn grandchild, conveying that unvaccinated adults are a mortal threat to infants. Plaintiff Lori DeCostanzo alleged that GSK’s advertising falsely implied that Boostrix would prevent recipients from becoming infected with and transmitting pertussis bacteria, when in fact acellular pertussis vaccines are known to reduce symptoms in the vaccinated person without reliably blocking asymptomatic colonization or onward transmission. A New York district court denied GSK’s motion to dismiss in 2022, finding it plausible that GSK had a “special relationship” duty to give consumers correct information and that the plaintiff had suffered a cognizable injury. GSK settled the case in 2026 without admitting wrongdoing, offering class members $10 to $50 in compensation. The settlement notice explicitly clarified that the plaintiff did not challenge the safety or efficacy of Boostrix in protecting vaccine recipients; the case was solely about the transmission-blocking claims that underpinned the cocooning marketing strategy. That distinction is precisely the one the scientific literature had already drawn: acellular pertussis vaccines protect the recipient but do not reliably protect others.
Bottom line on cocooning:The strategy is theoretically reasonable but practically undeliverable at the required coverage levels and economically indefensible as a primary infant-protection measure in low-incidence settings. Its continued recommendation as a supplement is defensible only if the costs to individuals and healthcare systems are acknowledged honestly.
STRATEGY TWO
Maternal Vaccination: Better Evidence, but Read the Small Print
Maternal Tdap vaccination during the third trimester of pregnancy is a fundamentally different strategy. When a pregnant woman receives the vaccine between 27 and 36 weeks of gestation, her immune system produces pertussis antibodies that cross the placenta and enter the fetus. The baby is born already carrying its mother’s immunity, protecting it from day one, before its own vaccination series begins at two months.
The evidence for this strategy is meaningfully stronger than for cocooning. Multiple large studies from the United States, United Kingdom, and Wales have found it effective. It is logistically simpler, requiring vaccination of one person at a predictable antenatal visit rather than a household network of uncertain composition. And it protects the infant not just from household contacts but from any source of infection, because the protection is conferred directly on the baby.
All of this is true. But the way effectiveness is communicated by government agencies and medical bodies deserves scrutiny.
The Headline vs. the Reality
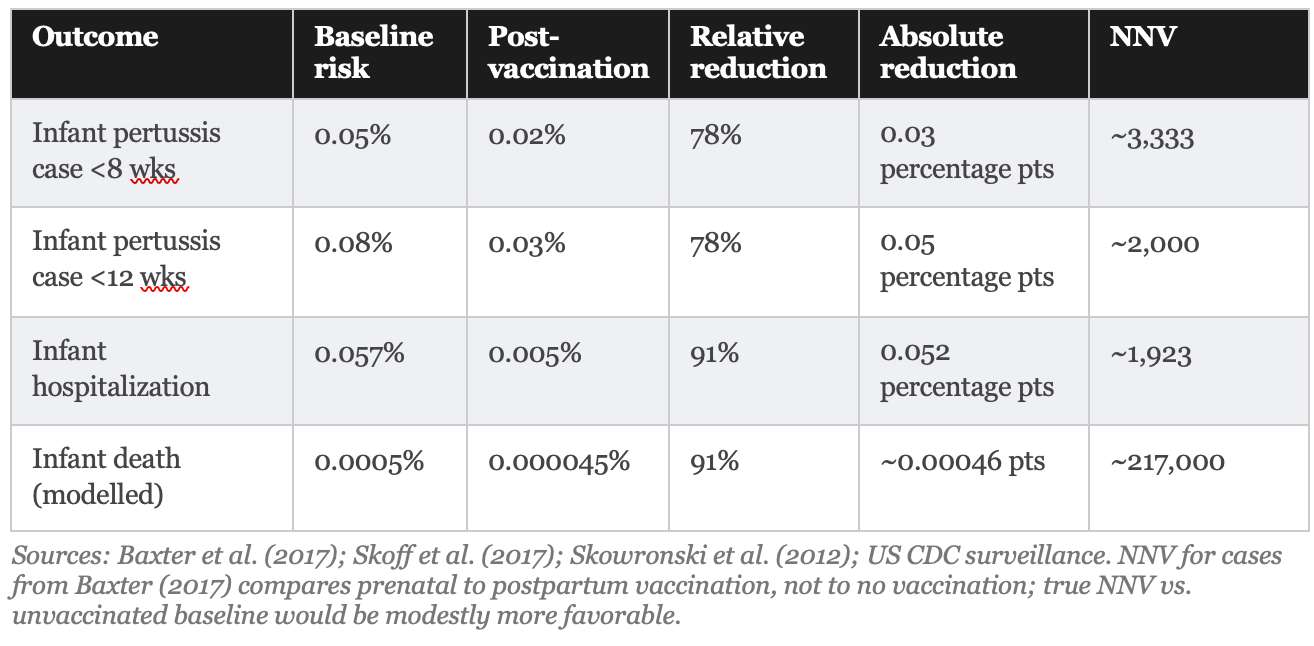
Official communications uniformly report relative effectiveness: the vaccine is “78% effective” at preventing pertussis cases in infants under 2 months and “91% effective” against hospitalization. These figures, derived from high-quality studies, are accurate. They are also systematically incomplete without the corresponding absolute figures.
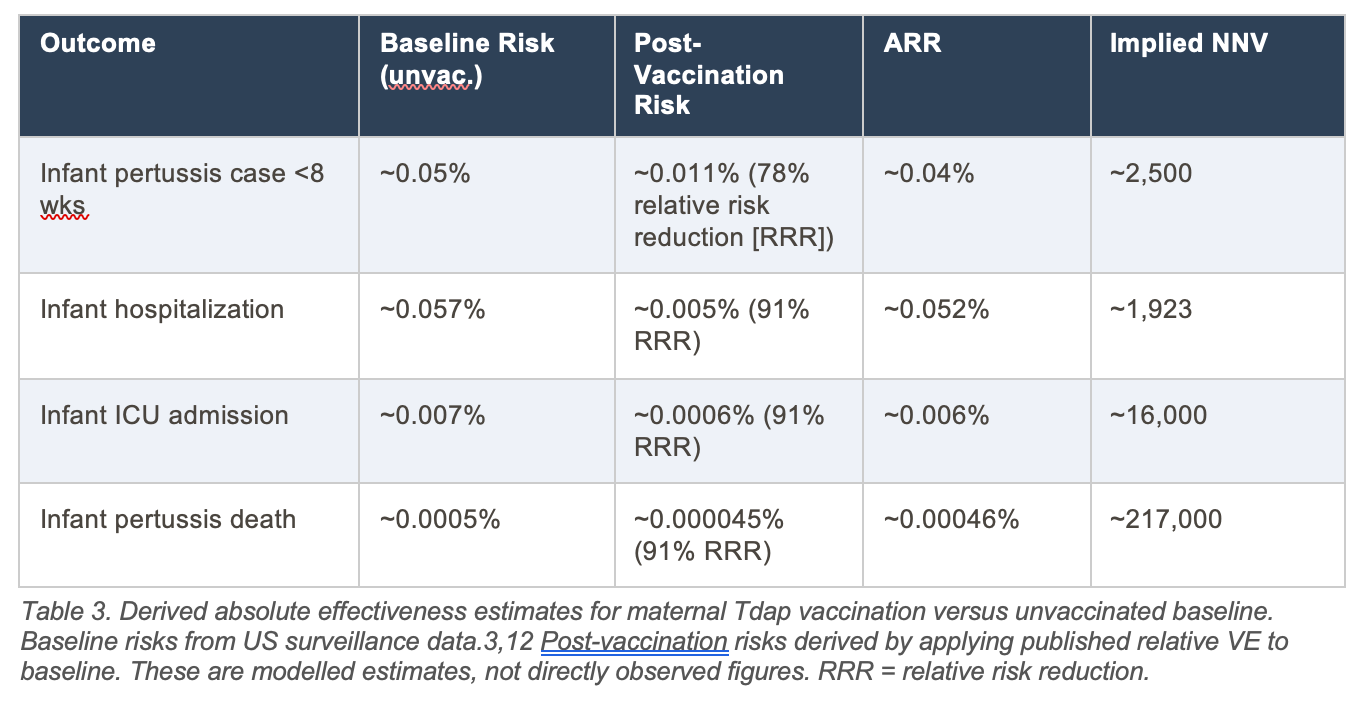
A relative risk reduction tells you how much smaller the risk becomes as a percentage. An absolute risk reduction tells you how many fewer people actually experience the outcome. When baseline risk is low, as it is for infant pertussis in the contemporary United States, even large relative reductions correspond to small absolute changes. This is not a flaw in the vaccine. It is a mathematical reality that belongs in every patient conversation.
What these numbers mean in practical terms: if you vaccinate 3,333 pregnant women who would otherwise be vaccinated postpartum, you prevent approximately one infant pertussis case in the first eight weeks. Vaccinating 217,000 pregnant women prevents approximately 1 infant death from pertussis. These are population-level benefits, not individual-level guarantees, and they should be communicated as such.
Patients told 'this vaccine prevents 91% of hospitalized cases” and patients told ‘vaccinating 1,923 pregnant women prevents one hospitalization’ are receiving the same information in very different forms. The first framing produces compliance. The second produces informed consent.
THE COSTS AND RISKS
The economic picture for maternal vaccination is substantially more favorable than for cocooning, but still requires honest presentation. Recent US decision-analytic models estimate strategy costs ranging from $7,601 to $414,523 per QALY saved, depending heavily on model assumptions and on whether the benefit of protecting the vaccinated mother herself is included.
The lower figure ($7,601/QALY) comes from analyses that count both infant protection and maternal protection as benefits. The higher figure ($414,523/QALY, exceeding the standard US threshold of $100,000–$159,429/QALY) comes from models that evaluate the strategy solely for infant protection, the stated purpose of the recommendation. The difference is not trivial. Policymakers and patients should know which framing underlies the cost-effectiveness claim.
The estimated cost per infant death prevented (the most emotionally resonant outcome) is approximately $14 million to $22 million for maternal vaccination and >$65 million to $100 million for cocooning. These are order-of-magnitude estimates based on US vaccine costs ($65–$100 per dose, including administration) and modeled NNV figures. They are not small numbers, and they are almost never disclosed in public health communications about these programs.
The Safety Signal That Should Be on the Consent Form
One large observational study (Layton et al., 2017) following over one million pregnancies found a small statistical association between maternal Tdap and two adverse outcomes that are rarely mentioned in patient-facing materials:
Chorioamnionitis (inflammation of the fetal membranes): 3.3% in vaccinated women versus 3.0% in unvaccinated women. Absolute risk difference: 0.3 percentage points. Implied number needed to harm (NNH): approximately 333.
Postpartum hemorrhage: 2.9% in vaccinated versus 2.4% in unvaccinated. Absolute risk difference: 0.5 percentage points. Implied NNH: approximately 200.
IMPORTANT CONTEXT: These signals have not been replicated in randomized controlled trials. Multiple independent studies, including a large British national program evaluation, have not confirmed them. The most plausible explanation is detection bias: vaccinated women have more prenatal healthcare contact and are therefore more likely to have these conditions diagnosed. The Canadian regulatory review concluded there is no confirmed increased risk. The signals are probably not real causal effects. But they should be disclosed to patients regardless, because patients cannot make informed decisions about information they are not given.
If the chorioamnionitis signal were confirmed as causal, the NNV-to-NNH calculation becomes uncomfortable for the mortality endpoint specifically: NNV of 217,000 to prevent one infant death, versus NNH of 333 to cause one chorioamnionitis case. That ratio (652:1 in favor of harm for those two endpoints) does not mean the vaccine is harmful overall; chorioamnionitis is not equivalent to infant death. But it is precisely the kind of number that informs risk-benefit conversations, and it belongs in those conversations.
THE COMPARISON
Head to Head: What the Evidence Actually Shows
THE BOTTOM LINE
What an Objective Reading of the Evidence Requires Us to Acknowledge
An objective reading of the public health evidence on pertussis vaccination does not lead to the conclusion that these vaccines are harmful or should be avoided. It leads to something more specific: that the institutional communication of their benefits has been systematically incomplete in ways that undermine genuine informed consent.
The review article that is the basis for this summary makes four observations that a rigorous and honest public health system would elevate rather than minimize:
1. Cocooning has never worked. The strategy has been known to be insufficient since at least 2011. It has never achieved the coverage required to approach its theoretical potential. Yet it remains on official CDC and ACIP recommendation lists without the prominent caveat that its cost-effectiveness is indefensible at low pertussis incidence. Recommending a strategy that costs an estimated $65 million to $100 million per infant death prevented without disclosing that figure is not a neutral communication choice.
2. Relative effectiveness is not the same as absolute effectiveness. A vaccine that is “91% effective” against infant pertussis hospitalization reduces the absolute risk from approximately 0.057% to 0.005%. Both figures are true. Only one of them, the absolute figure, is useful for a parent trying to weigh the intervention against alternatives and risks. Government agencies (and pharmaceutical marketing campaigns) are consistently aligned in leading with the relative figure and omitting the absolute one.
3. The chorioamnionitis signal should be on the consent form. The signal may well be a false positive caused by detection bias. The review article says as much. But the appropriate response to an unconfirmed adverse signal is not to omit it from patient counseling; it is to disclose it with appropriate contextualization. Patients are capable of understanding nuance. They should be trusted to do so.
4. Cost per outcome prevented matters. Telling policymakers that maternal Tdap is “cost-effective” without specifying whether that claim is based on a narrow payer perspective (where the cost per QALY can exceed the US threshold) or a broader societal perspective (where it may be cost-saving) is not a transparent analysis. It is advocacy dressed as science.
The appropriate question is not whether a vaccine is effective. The appropriate question is: effective at what, for whom, at what absolute magnitude, and at what cost and risk, compared to what alternative?
These are not anti-vaccine arguments. They are pro-transparency arguments. The distinction matters because conflating the two, namely treating any demand for complete data as an attack on vaccination, is itself a source of institutional credibility damage that the public health establishment can ill afford.
THE CONCLUSION
What This Review Actually Supports
The rigorous, objective comparative analysis that underlies this summary reaches a clear hierarchy:
Maternal Tdap vaccination during the third trimester is a better strategy relative to cocooning. It is more effective, more logistically tractable, and more cost-effective, even by conservative estimates, and it confers protection on the infant from any source of infection, rather than only from household contacts. The NNVs for preventing infant cases (~3,333) and hospitalizations (~1,923) fall within ranges most clinicians would consider reasonable for a safe vaccine with a favorable short-term safety profile. But mandates or strong-arming pregnant women to accept this option is completely inappropriate; proper informed consent and shared decision making should be standard clinical practice.
Cocooning policy advocacy by public health officials, obstetricians, or pediatricians should be retained only as a supplement. For infants whose mothers were not vaccinated during pregnancy, cocooning offers some incremental protection from household transmission. As a standalone strategy in a low-incidence setting, its NNV for serious outcomes and cost per QALY are not defensible without substantial transparency regarding those figures. The current CDC and ACIP position of broad endorsement of cocooning should be rescinded.
Both strategies should be communicated with absolute figures alongside relative ones. An NNV of 217,000 to prevent one infant death is not a reason to avoid vaccination. It is a reason to understand what each vaccination strategy is and what it is unlikely to achieve for the individual patient, and to make the decision accordingly.
The unresolved safety signals should be disclosed. Not as a reason to decline vaccination, but as a reason to have an honest conversation, the kind of conversation that public health authorities have historically been reluctant to facilitate because they fear it will reduce uptake. That fear is understandable. It is also a poor basis for informed consent.
The review article’s own conclusion: Of the two, maternal Tdap vaccination is the more appropriate primary strategy. The evidence for it is stronger than the evidence for cocooning. Its safety profile is broadly reassuring. And the honest disclosure of its absolute risk reductions, cost per outcome, and unresolved safety signals would, if anything, support rather than undermine a well-informed patient’s decision to receive it.
REFERENCE
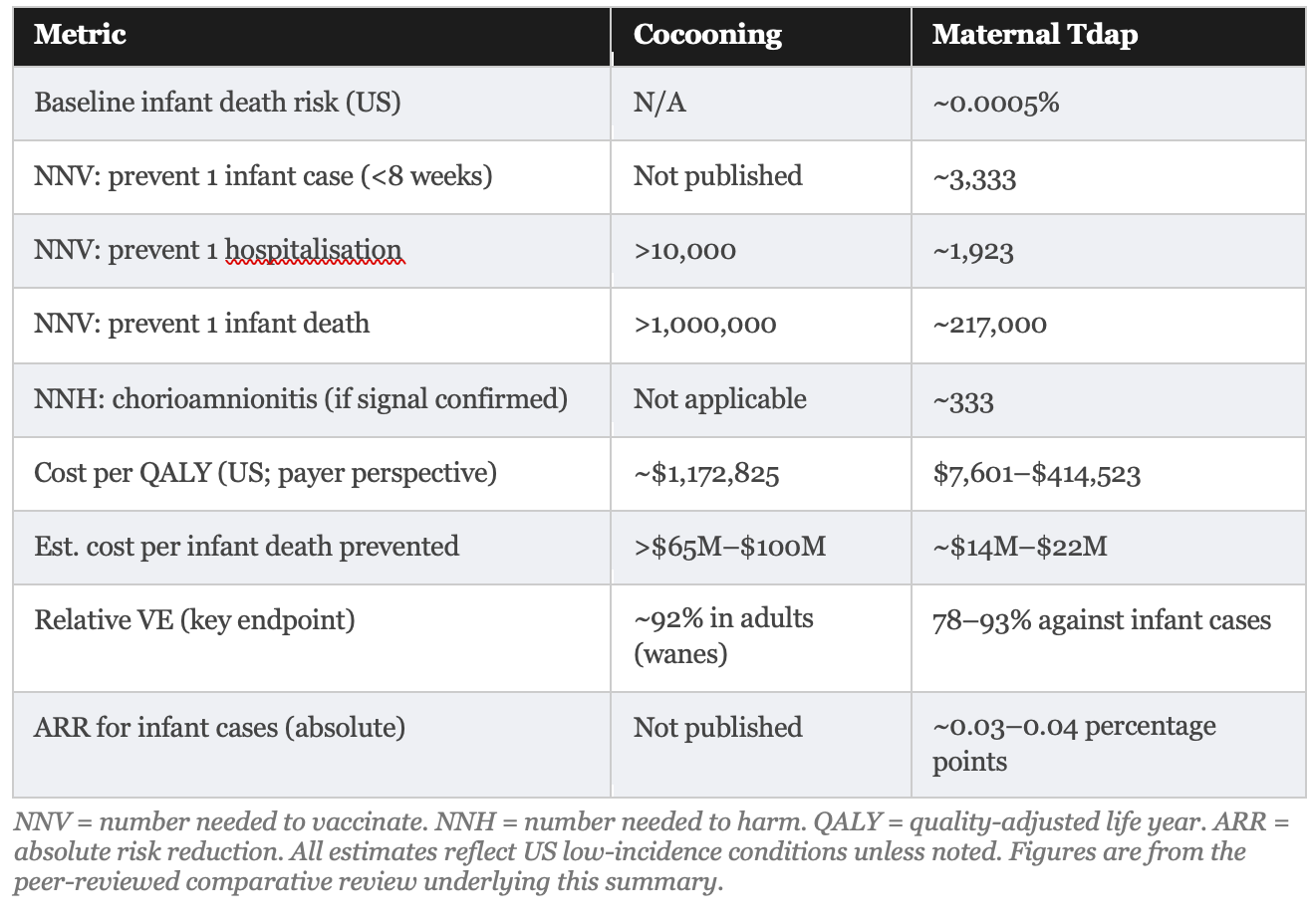
Key Numbers at a Glance
About this summary
This piece summarizes a detailed comparative review article on cocooning versus maternal Tdap vaccination to prevent pertussis in newborns. All numerical figures are derived from published studies cited in the source manuscript. NNV and cost estimates are epidemiological models based on observed baseline incidence rates and represent expected values rather than precise predictions for individual patients or populations. Local incidence conditions substantially affect NNV and cost-effectiveness figures. This summary does not constitute medical advice. Pregnant individuals with questions about Tdap vaccination should consult their obstetric provider.
Key sources
Skowronski DM et al. Clin Infect Dis. 2012;54(3):318–327
Baxter R et al. Pediatrics. 2017;139(5):e20164091
Skoff TH et al. Clin Infect Dis. 2017;65(12):1977–1983
Terranella A et al. Pediatrics. 2013;131(6):e1748–e1756
Hersh AR et al. Obstet Gynecol. 2023;141(3):547–556
Layton JB et al. Vaccine. 2017;35(33):4072–4078
Westra TA et al. Clin Ther. 2010;32(8):1479–1495
Fernandez-Cano MI et al. Vaccine. 2015;33(19):2213–2220
Munoz FM et al. JAMA. 2014;311(17):1760–1769
DeSilva M et al. Vaccine. 2017;35(36):4761–4769
CDC Manual for Surveillance of Vaccine-Preventable Diseases, Chapter 10 (2026)
DeCostanzo v. GlaxoSmithKline plc et al., No. 2:21-cv-04869-NJC-AYS (E.D.N.Y. 2020); settled March 2026
Cocooning Versus Maternal Tdap Vaccination During Pregnancy
A Critical Comparative Review of Risks and Benefits for Prevention of Pertussis Morbidity and Mortality in Newborns
Author: Robert W. Malone, MD, MS with support from Claude AI (Anthropic)
Background: Pertussis (whooping cough) remains a leading cause of vaccine-preventable infant mortality worldwide. Two strategies protect newborns before their own immunization can begin: (1) cocooning, whereby close contacts receive tetanus, diphtheria, and acellular pertussis (Tdap) vaccine after delivery, and (2) maternal Tdap vaccination during the third trimester of pregnancy, conferring passive immunity via transplacental antibody transfer. Despite broad endorsement of both approaches, the quantitative evidence supporting their respective risk-benefit profiles warrants careful and objective scrutiny, particularly with respect to absolute rather than relative measures of effectiveness and cost.
Abstract
Background: Pertussis (whooping cough) remains a leading cause of vaccine-preventable infant mortality worldwide. Two strategies protect newborns before their own immunization can begin: (1) cocooning, whereby close contacts receive tetanus, diphtheria, and acellular pertussis (Tdap) vaccine after delivery, and (2) maternal Tdap vaccination during the third trimester of pregnancy, conferring passive immunity via transplacental antibody transfer. Despite broad endorsement of both approaches, the quantitative evidence supporting their respective risk-benefit profiles warrants careful and objective scrutiny, particularly with respect to absolute rather than relative measures of effectiveness and cost.
Methods: This narrative review synthesizes published data on relative vaccine effectiveness (VE), absolute risk reduction (ARR), number needed to vaccinate (NNV), number needed to harm (NNH), cost-effectiveness, implementation challenges, and cost per outcome prevented for both strategies. Evidence is drawn from randomized controlled trials, observational cohort studies, pharmacovigilance databases, health economic modelling studies, and published cost-effectiveness analyses.
Results: The NNV for the cocooning strategy to prevent one infant death exceeds 1,000,000 under low-incidence conditions; the NNV to prevent one hospitalization exceeds 10,000. For maternal Tdap vaccination, the relative VE against infant pertussis cases within the first eight weeks of life is 78%, but this corresponds to an ARR of approximately 0.03 percentage points, yielding an NNV of approximately 3,333 compared with postpartum vaccination. Against infant hospitalization, relative VE is approximately 91%, ARR approximately 0.05%, and implied NNV approximately 1,923. Against infant death, the modelled NNV is approximately 217,000. Cost-effectiveness analyses place maternal Tdap at $7,601 to $414,523 per quality-adjusted life year (QALY) in United States analyses versus $1,172,825 per QALY for cocooning. The estimated cost per infant death prevented is $14 million to $22 million for maternal vaccination versus $65 million to $100 million for cocooning. No formal NNH for serious, causally confirmed harm from either strategy has been established.
Conclusions: Maternal Tdap vaccination during pregnancy is the more effective, logistically tractable, and cost-effective strategy, with a cost per QALY of $7,601 to $414,523 versus $1,172,825 for cocooning in United States analyses. However, the gap between relative VE figures (78–93%) and modest absolute risk reductions (ARR 0.03–0.05% for cases and hospitalization; estimated cost per infant death prevented of $14 million to $22 million) reflects the low background incidence of severe infant pertussis in high-coverage settings. Clinicians and policymakers should present absolute alongside relative effectiveness and cost data when counselling patients.
Keywords: pertussis; Tdap; cocooning; maternal vaccination; absolute risk reduction; number needed to vaccinate; relative versus absolute effectiveness; cost-effectiveness; cost per QALY; whooping cough; newborn protection
1. Introduction
Pertussis (whooping cough) is caused by Bordetella pertussis, an obligate human pathogen transmitted via respiratory droplets. Despite decades of childhood immunization, pertussis has not been eliminated and the World Health Organization (WHO) estimates 20 to 40 million cases annually worldwide, with approximately 160,700 deaths in children under five years of age documented in 2014.1,2
Severe disease and death are concentrated disproportionately in infants younger than three months of age, who are too young to have completed their primary immunization series. In the United States (US), from 2013 through 2022, 57% of all pertussis-related deaths occurred in infants younger than two months.3
Two principal strategies attempt to close this immunity gap. The cocooning strategy vaccinates all close contacts of the newborn after delivery, reducing household transmission. Maternal vaccination during the third trimester generates pertussis-specific antibodies that cross the placenta, providing the infant with humoral immunity from birth.4
Both strategies are endorsed by the CDC Advisory Committee on Immunization Practices (ACIP), the American College of Obstetricians and Gynecologists (ACOG), and the WHO. However, public health messaging for both strategies has overwhelmingly reported relative effectiveness, a practice that can mislead clinicians and patients about the absolute magnitude of individual benefit and cost. This review provides a direct comparative analysis using both relative and absolute effectiveness metrics, NNV, NNH, and cost-effectiveness data.
2. Background: Pertussis Epidemiology and Baseline Risks
2.1 Pertussis Incidence and the Absolute Baseline
Understanding absolute effectiveness requires knowing the baseline risk of each outcome in the absence of the intervention. For infant pertussis in the United States, overall infant pertussis incidence varies between approximately 55 and 200 per 100,000 in non-epidemic and epidemic years respectively. Pertussis-related infant mortality in the current US surveillance era is approximately 0.1 to 0.5 per 100,000 live births per year (0.0001% to 0.0005% per birth), reflecting both the low incidence of fatal cases and the protective effect of existing vaccination programs.3,6
These baseline figures are the denominator against which all absolute effectiveness calculations must be performed. Their magnitude has direct implications for NNV estimates, cost-effectiveness ratios, and for the interpretation of relative VE figures.
2.2 Resurgence, Waning Immunity, and the Rationale for Supplemental Strategies
The resurgence of pertussis in vaccinated populations reflects several factors: faster waning of immunity from acellular pertussis (aP) compared with whole-cell (wP) vaccines, possible antigenic mismatch with emerging pertactin-deficient strains, improved diagnostic sensitivity, and under-diagnosis in prior decades.8,9
Household and close contacts account for 35% to 75% of infant infections, with the mother being the most commonly identified source. However, approximately 34% of infant infections are attributed to casual contacts outside the household, a source that cocooning cannot address.4,10
3. The Cocooning Strategy
3.1 Mechanism and Theoretical Basis
Formally endorsed by the CDC ACIP beginning in 2005, cocooning involves administering Tdap to all individuals likely to have close contact with a newborn who have not previously received an adult Tdap dose.11
3.2 Relative Vaccine Effectiveness in Adults
Relative VE of Tdap in adults against typical pertussis disease has been reported at approximately 92% in a double-blind randomised controlled trial (RCT), though with a wide confidence interval (CI): 95% CI 33% to 99%.4
This relative VE wanes rapidly. Studies of adolescents show Tdap providing approximately 69% relative protection within the first year, declining to less than 9% after four years.8
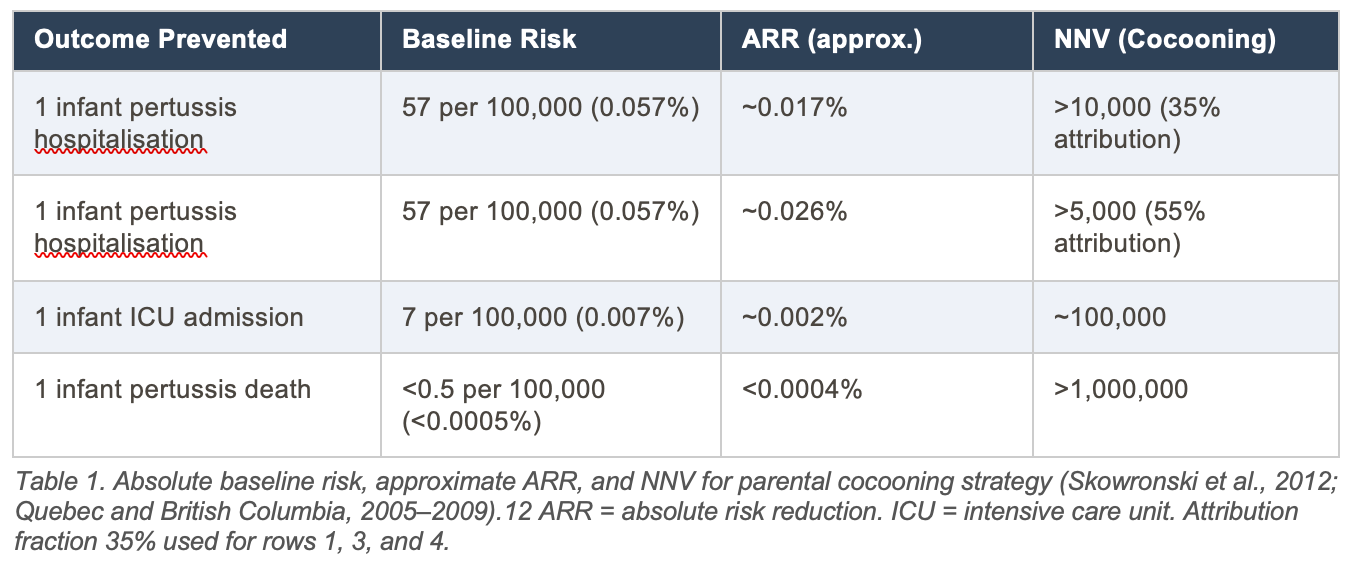
3.3 Absolute Effectiveness and NNV for Cocooning
The most rigorous published NNV analysis for parental cocooning is that of Skowronski and colleagues (2012), using Canadian surveillance data from Quebec and British Columbia (2005–2009). Their formula was: NNV = 2 parents / (parent-attributable infant risk x parent vaccine efficacy), with parental attribution set at 35% (explored to 55%) and adult VE at 85%.12
Interpretive note: The NNV for infant death via cocooning exceeding 1,000,000 reflects the product of a low absolute baseline mortality risk (<0.0005%) and a strategy that addresses only the parental fraction of transmission (35–55%). The relative VE of 85% in adults appears high, but its effect on the absolute infant mortality rate is negligible at this baseline.
3.4 Implementation Challenges
Real-world cocooning coverage consistently falls below the 92% to 94% threshold estimated to be necessary for meaningful herd protection. A Swiss cross-sectional study found vaccination rates of 61% to 69% in mothers and 58% to 59% in fathers, with rates dropping to 18% for uncles, 13% for aunts, and 7% for grandparents.14
The CDC ACIP concluded in 2011 that cocooning alone was insufficient to prevent pertussis morbidity and mortality in newborns.11
3.5 Safety Profile in Adults
The most common adverse effects of Tdap in adults are local injection-site reactions in the majority of recipients, and mild transient systemic reactions. No NNH for a serious, causally established harm from Tdap in adults has been published. The Vaccine Safety Datalink found a medically attended event rate of 8.1 per 10,000 within three days of vaccination in pregnant women, not statistically different from the unvaccinated control rate of 6.8 per 10,000 (adjusted incidence rate ratio [AIRR] 1.19; 95% CI: 0.81 to 1.73).15
4. Maternal Tdap Vaccination During Pregnancy
4.1 Mechanism
Maternal immunoglobulin G (IgG) is actively transported across the placenta from approximately 28 to 32 weeks of gestation. Infants born to mothers vaccinated during the third trimester carry higher concentrations of pertussis-specific antibodies at birth. Optimal timing is between 27 and 36 weeks’ gestation, as early as possible in that window.16,17
One caveat is immune interference. High maternally derived antibody concentrations may modestly blunt the infant’s own immune response to the primary diphtheria, tetanus, and acellular pertussis (DTaP) series. Randomized trial data show lower pertussis antibody concentrations after the third DTaP dose in infants of vaccinated mothers, though differences generally disappear after the fourth dose.18
4.2 Relative Vaccine Effectiveness
A Centers for Disease Control and Prevention (CDC) case-control evaluation found maternal Tdap between 27 and 36 weeks to be 78% effective (relative) in preventing pertussis cases in infants younger than two months, and 91% effective against hospitalised pertussis cases.19
A large British national program cohort study reported 91% relative VE in infants younger than three months. A Welsh case-control study found adjusted relative VE of 93% (95% CI: 81% to 97%) in infants younger than eight weeks.20
These are relative measures. To understand their clinical significance for individual patients, they must be translated into absolute terms using baseline risk data. This is where the review performs its most important service: separating what the numbers say from what they mean.
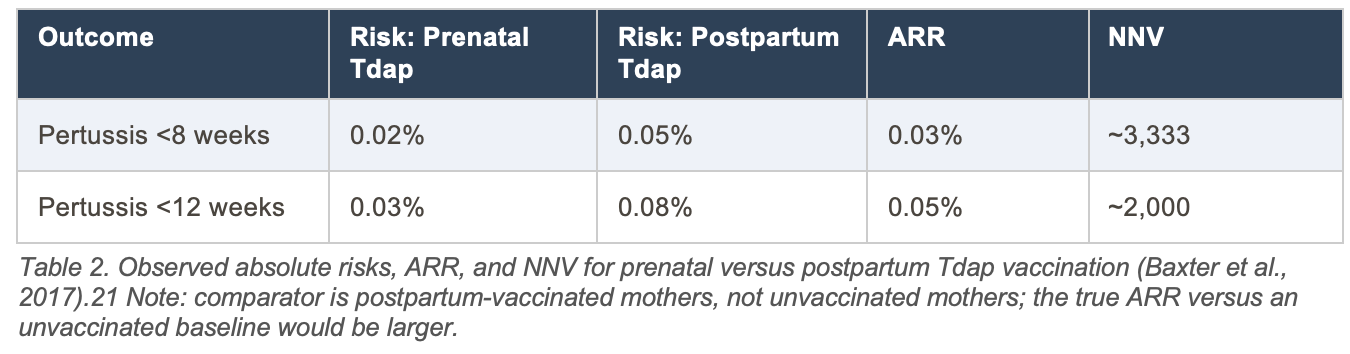
4.3 Absolute Effectiveness and NNV for Maternal Vaccination
The 2017 California cohort study by Baxter and colleagues reported observed risks in both the vaccinated and comparator groups, making direct ARR calculation possible.21
Interpretive note on infant mortality NNV: The modelled NNV of ~217,000 to prevent one infant death reflects an extremely low absolute baseline mortality risk (~0.0005% per birth). Even a highly effective intervention (91% relative VE) produces a small ARR at this baseline. No randomized trial has been powered to detect a mortality difference for this reason.
4.4 The Interpretive Gap Between Relative and Absolute Effectiveness
The near-universal reporting of relative VE in the pertussis vaccine literature has consequences here. A relative VE of 78% or 91% against infant pertussis cases or hospitalization sounds clinically compelling. The corresponding ARRs of approximately 0.03% to 0.05% for cases and hospitalization, and 0.00046% for death, convey a different sense of individual-level benefit. Both figures are mathematically consistent and epidemiologically valid; the choice of which to emphasize is not neutral.5
4.5 Comparative Effectiveness: Maternal Vaccination Versus Cocooning
Using a cohort decision model, Terranella and colleagues compared infant protection strategies and found that vaccination during pregnancy prevented a higher proportion of cases (33% vs. 20%) and deaths (49% vs. 16%) than cocooning, at a lower cost per quality-adjusted life year (QALY). The structural advantages of maternal vaccination are detailed further in the cost-effectiveness analysis (Section 7); in brief, (1) it confers direct infant immunity rather than reducing only household transmission, so it addresses casual-contact infections as well; (2) it requires vaccination of only one individual at a predictable healthcare encounter; and (3) its efficacy does not depend on achieving herd-immunity-level coverage across an extended household network.4
4.6 Safety Profile
4.6.1 Randomized Controlled Trial Evidence
The randomized controlled trial (RCT) by Munoz and colleagues found no Tdap-associated serious adverse events in vaccinated women or their infants. Injection site reactions were reported in 78.8% of pregnant women. Systemic reactions were reported in 36.4% of pregnant compared with 73.3% of postpartum women.18
Four additional RCTs comparing Tdap versus placebo or tetanus toxoid in pregnancy reported no differences in adverse outcome rates and no serious vaccine-related adverse events.22
4.6.2 Large Cohort Studies
The Vaccine Safety Datalink cohort of 53,885 vaccinated and 109,253 matched unvaccinated pregnant women found no statistically significant increase in medically attended composite acute adverse events within three days of vaccination (AIRR 1.19; 95% CI: 0.81 to 1.73). The only statistically elevated signal was medically attended fever: 2.8 per 10,000 vaccinated versus fewer than 1 per 10,000 controls (AIRR 5.4; 95% CI: 2.1 to 13.9), with low absolute rates.15
4.6.3 The Chorioamnionitis Signal
One large observational study by Layton and colleagues (n=1,079,034 pregnancies) found a small association between optimally timed maternal Tdap and chorioamnionitis (3.3% vaccinated versus 3.0% unvaccinated; relative risk [RR] 1.11; 95% CI: 1.07 to 1.15) and postpartum hemorrhage (2.9% versus 2.4%; RR 1.23).23
The absolute risk differences are small: 0.3 percentage points for chorioamnionitis and 0.5 percentage points for postpartum hemorrhage, implying NNH values of approximately 333 and 200, respectively. However, the observational design is subject to confounding by healthcare contact intensity. Neither signal has been replicated in RCT data or confirmed by Canadian regulatory review, which concluded no increased risk of serious adverse events has been found.22
If the chorioamnionitis signal were confirmed as causal, the NNV-to-NNH ratio for cases would be approximately 3,333:333 = 10:1 in favor of vaccination benefit. For infant death, the NNV of ~217,000 compared with an NNH of ~333 would represent a ratio of approximately 652:1 against vaccination for those two specific endpoints. That arithmetic is worth being honest about.
4.6.4 Vaccine Adverse Event Reporting System (VAERS) Surveillance
A 20-year pharmacovigilance analysis of VAERS data (2005–2024) identified 870 pregnancy-related reports from 20,358 Tdap submissions, of which 128 were classified as serious. No maternal deaths were reported. VAERS cannot calculate true incidence or NNH due to passive surveillance limitations.24
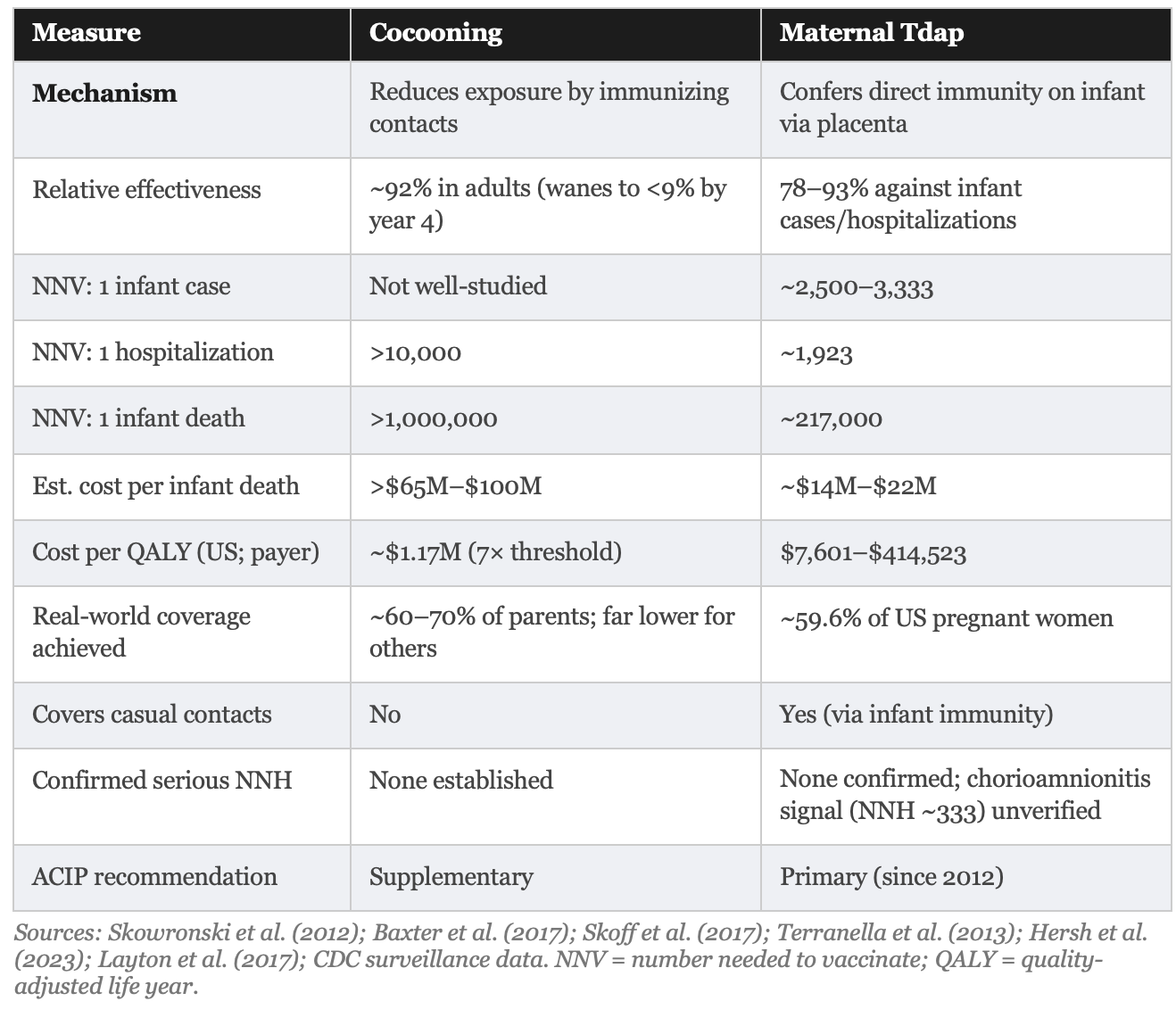
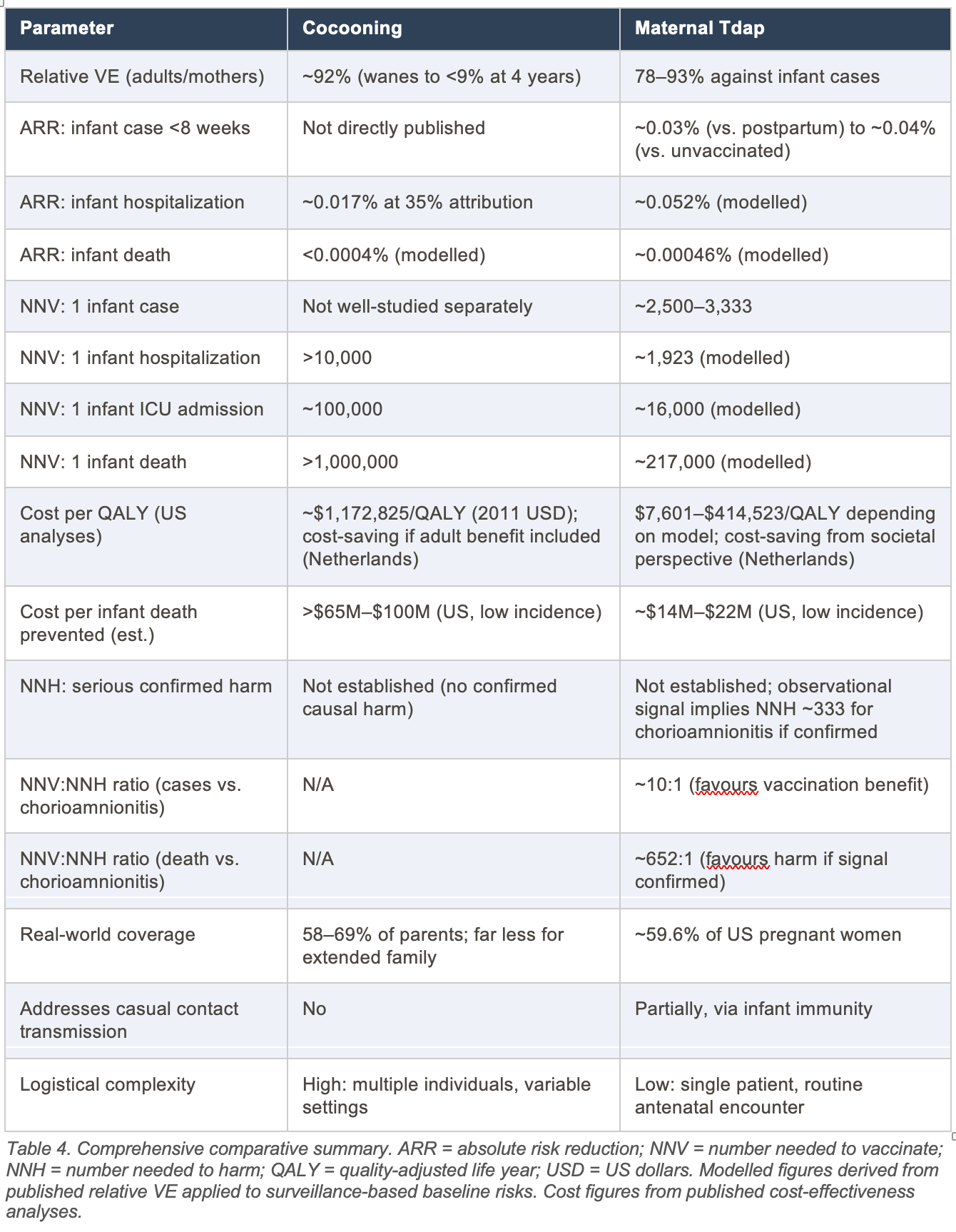
5. Comprehensive Comparative Summary
6. Critical Appraisal of the Evidence
6.1 The Relative vs. Absolute Effectiveness Problem
The near-universal reporting of relative VE in the pertussis vaccine literature has particular consequences. A relative VE of 78% or 91% against infant pertussis cases or hospitalization sounds clinically compelling. The corresponding ARRs of approximately 0.03% to 0.05% for cases and hospitalization, and 0.00046% for death, convey a different sense of individual-level benefit. Public health recommendations that rely primarily on relative VE may be contributing to a systematic overestimation of individual benefit among clinicians and patients.5
6.2 Methodological Limitations Shared by Both Strategies
NNV estimates are highly sensitive to the background incidence rates used as input. All figures derived from low-incidence surveillance periods will appear unfavorable. The primary evidence base for maternal vaccination efficacy consists largely of observational studies. No randomised trial has been powered to detect differences in the rare outcome of infant death, meaning NNV estimates for that endpoint are necessarily modelled rather than observed, with substantial uncertainty.
6.3 The Mortality Endpoint Requires Special Caution
Infant pertussis mortality in high-income settings is so rare that the absolute benefit of either vaccination strategy against this specific outcome is almost unmeasurably small in any single population study. The NNV estimates for preventing one infant death (>1,000,000 for cocooning; ~217,000 for maternal Tdap) and the corresponding cost estimates ($65M–$100M and $14M–$22M respectively) reflect genuine epidemiological reality, not a deficiency of the vaccines. The appropriate framing is not that these strategies are ineffective, but that they should not be justified primarily on mortality grounds when presenting to patients in low-incidence settings.
6.4 The Chorioamnionitis NNH and Its Uncertainty
The observational chorioamnionitis and postpartum hemorrhage signals from the Layton study, if confirmed as causal, would substantially change the NNV:NNH balance for the infant mortality endpoint specifically, where NNV (~217,000) exceeds NNH (~333) by approximately 650-fold. This would represent net harm. However, the causal status of this signal remains unresolved and it was not replicated in RCT data. The appropriate response is ongoing active surveillance, not withdrawal of the recommendation, but the signal should be disclosed transparently to patients.
6.5 Coverage Gaps Limit Population-Level Impact
Both strategies face persistent coverage gaps in real-world implementation. With maternal vaccination coverage at approximately 59.6% in the US, and cocooning coverage well below the herd-immunity threshold, neither strategy is achieving its theoretical potential at the population level.19,14
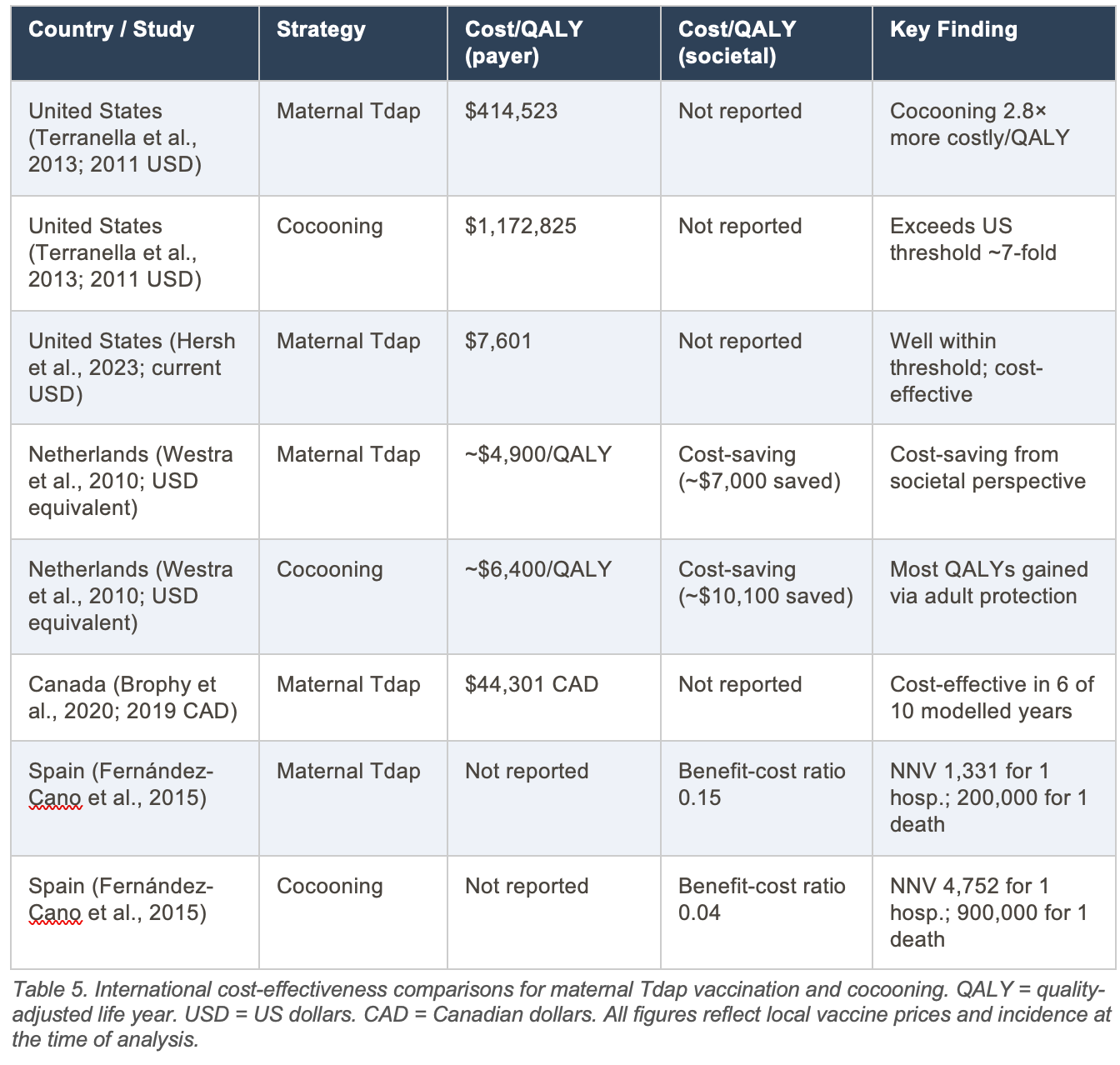
7. Cost-Effectiveness Analysis
7.1 Overview and Methodological Considerations
Cost-effectiveness analysis (CEA) of both vaccination strategies has been performed in multiple countries using decision-analytic models. The dominant metric is cost per quality-adjusted life year (QALY) gained, with a widely used United States threshold of approximately $100,000 to $159,429 per QALY considered cost-effective. All figures cited below are expressed in the year reported by the source study.27,28,29
Three factors dominate cost-effectiveness results across all published analyses: (1) the vaccine acquisition price, which varies substantially between healthcare systems; (2) the background incidence of pertussis at the time of analysis; and (3) the analytic perspective adopted, with societal-perspective analyses consistently yielding more favorable results than payer-perspective analyses because they capture productivity losses and caregiver time costs associated with infant and parental pertussis illness.
7.2 Cost-Effectiveness of Maternal Tdap Vaccination (United States)
The most comprehensive recent United States analysis, using a decision-analytic model applied to a theoretical cohort of 3.66 million annual pregnancies, found that universal maternal Tdap vaccination was cost-effective at $7,601 per QALY gained, using a vaccine cost of $47.75 per dose. The model estimated 22 fewer infant deaths, 11 fewer infant encephalopathy cases, 2,018 fewer infant hospitalizations, and 6,164 fewer infant pertussis infections per year.27
A second United States analysis using a vaccine cost of $41.19 found a cost per QALY of $6,481, also well within accepted thresholds. Sensitivity analyses confirmed the strategy remained cost-effective unless vaccine cost exceeded approximately $540 per dose, or unless maternal pertussis incidence fell below 1.6 cases per 10,000 pregnancies.28
From these parameters, the implied cost per outcome prevented can be estimated. With approximately 3.66 million vaccinations per year at a total cost of approximately $65 to $100 per dose (vaccine plus administration), the annual program cost is roughly $238 million to $366 million. Divided across 6,164 infant cases prevented, this yields an approximate cost per infant infection prevented of $38,600 to $59,400. For the 22 deaths prevented, the implied cost per infant death prevented is approximately $10.8 million to $16.6 million. These are model-derived estimates and reflect specific epidemiological assumptions.
7.3 Cost-Effectiveness of Cocooning (United States)
The Terranella and colleagues’ decision analysis comparing pregnancy vaccination with postpartum cocooning in the United States found the cost per QALY for pregnancy vaccination was substantially lower than for cocooning: $414,523 versus $1,172,825 per QALY respectively, both in 2011 US dollars. This 2.8-fold difference directly reflects the compounding inefficiencies of the cocooning strategy: the requirement to vaccinate multiple individuals, lower real-world coverage, and inability to address casual-contact transmission.35
Notably, the Terranella cost per QALY for maternal vaccination ($414,523) exceeds the standard US threshold of $100,000 to $159,429 per QALY. This finding illustrates the tension between cost-effectiveness from a narrow payer perspective and broader public health justification when the outcome being prevented (infant death) is rare in absolute terms. The more recent analyses yielding $7,601/QALY include protection of the vaccinated mother herself, producing a substantially more favorable ratio.35
Interpretive note: The discrepancy between the Terranella analysis ($414,523/QALY for maternal vaccination) and the more recent OBGYN analyses ($6,481–$7,601/QALY) reflects differences in model structure, epidemiological inputs, and whether maternal pertussis cases are counted as a benefit. Analyses that include protection of the vaccinated mother yield substantially more favourable cost-effectiveness ratios. The more conservative figure is arguably more appropriate when evaluating the strategy specifically for infant protection alone.
7.4 International Cost-Effectiveness Comparisons
The Netherlands analysis is the only major study to find both strategies cost-saving from a societal perspective. This result is driven primarily by the value assigned to preventing parental pertussis illness, rather than infant outcomes alone. When adults vaccinated under the cocooning program benefit personally from protection against pertussis infection, the strategy accumulates QALY gains that offset its cost. This methodological point is critical: cost-effectiveness of cocooning improves considerably when the vaccinated adult’s own health benefit is included.30
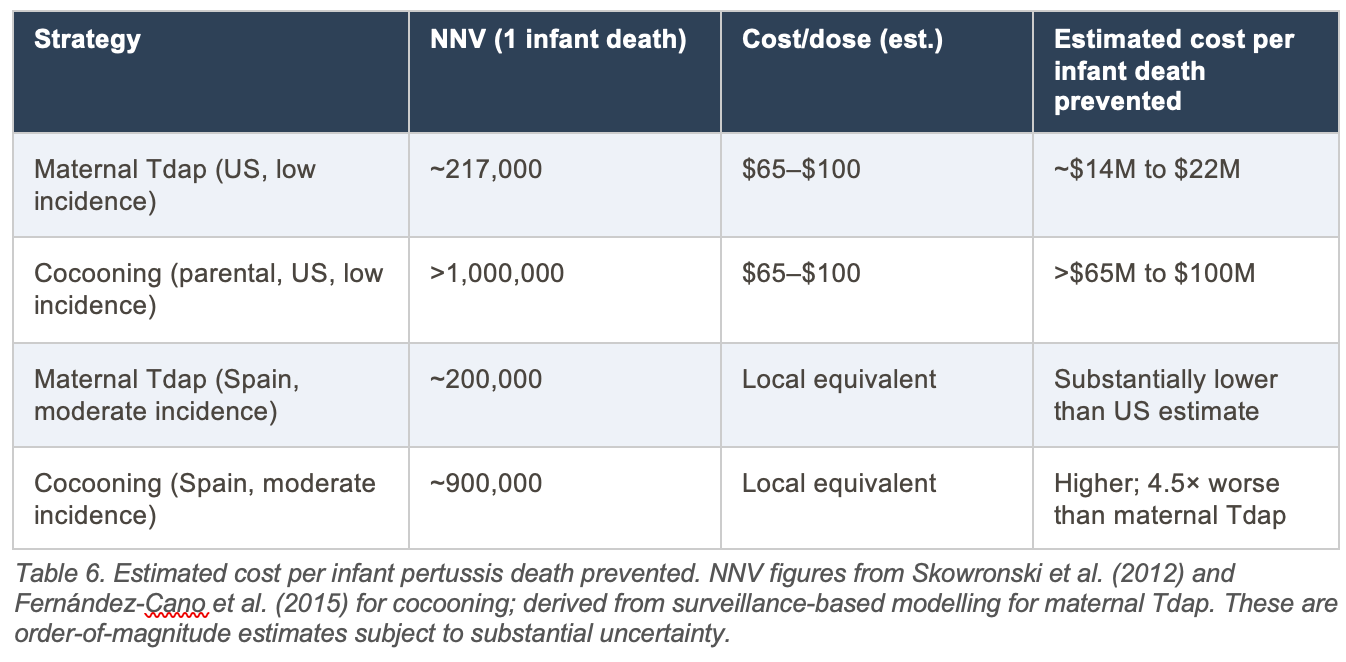
7.5 Cost Per Infant Death Prevented: A Direct Comparison
The cost per infant death prevented represents the most clinically salient economic metric. The following estimates are derived by multiplying NNV figures by approximate US vaccination costs ($65 to $100 per person including vaccine and administration).
Interpretive note: The estimated cost of $14M to $22M to prevent one infant death via maternal Tdap, and $65M to $100M via cocooning, may appear high in isolation. Context matters: US regulatory frameworks accept healthcare interventions costing $1M to $10M per life saved in other settings (e.g. neonatal intensive care). These pertussis figures are above typical QALY thresholds specifically because infant pertussis death is rare and per-dose cost is non-trivial. They do not indicate the strategies are without value, but they caution against framing vaccination programs primarily around mortality prevention when presenting individual-level benefit.
7.6 The Societal Perspective and the Case for Cocooning Rehabilitation
A consistent finding across the international literature is that the cost-effectiveness of both strategies improves substantially when the analysis shifts from a healthcare payer perspective to a societal perspective. The Dutch analysis finding both strategies to be cost-saving from a societal perspective, driven by preventing parental illness and associated productivity losses, suggests that narrow payer-perspective analyses may systematically undervalue these programs.30
Even from a societal perspective, maternal vaccination consistently yields a better cost-effectiveness ratio than cocooning in all jurisdictions studied, while also offering superior infant protection through direct immunological transfer. The economic evidence therefore reinforces rather than challenges the clinical and epidemiological case for prioritising maternal vaccination.
8. Recommendations for Clinicians and Policymakers
1. Maternal Tdap vaccination between 27 and 36 weeks of gestation is the more evidence-supported strategy of the two, offering a lower NNV for cases and hospitalisation, logistical tractability, and protection against both household and casual-contact transmission. It should be considered as the primary recommendation for all unvaccinated pregnant women, but should be communicated in the context of shared decision making between clinician and patient, with full disclosure of risks and potential benefits in the context of an informed consent process that is free of any form of coercion.
2. Cocooning remains a potential but problematic supplement for consideration, restricted to infants whose mothers did not receive antenatal vaccination, recognizing the challenge of implementation in most if not all real-world situations. Its role should be explicitly framed as supplementary rather than primary, given the very high NNV for serious infant outcomes and consistently suboptimal real-world coverage.
3. CDC ACIP policy should be revised to reflect these findings, and the current policy recommendation supporting cocooning should be rescinded.
4. Clinicians should present both relative and absolute effectiveness figures during patient counseling. A statement such as: “This vaccine reduces your baby’s risk of pertussis in the first eight weeks by about 78%, which means the absolute risk falls from roughly 5 in 10,000 to roughly 1 in 10,000” is more informative than relative VE alone and supports genuinely informed consent.
5. The unconfirmed chorioamnionitis and postpartum haemorrhage signals from observational data should be disclosed to patients with appropriate contextualisation: the signals are not confirmed in RCT data, the most plausible explanation is detection bias, and the NNV:NNH ratio for the case-prevention endpoint remains favourable even if the signal were confirmed.
6. Policymakers should evaluate NNV and cost estimates against local epidemiological conditions. In high-incidence settings or during outbreak periods, NNV figures improve materially and the case for universal vaccination strengthens. In low-incidence settings, the efficiency of universal versus risk-stratified approaches deserves explicit analysis.
7. Cost-effectiveness analyses consistently support maternal Tdap vaccination over cocooning as the more efficient use of program resources, but neither option is economically compelling. At current United States vaccine prices, maternal vaccination costs approximately $7,601 per QALY gained in recent analyses, well within the standard $100,000 to $159,429 per QALY threshold. Cocooning costs approximately 2.8 times more per QALY than maternal vaccination in comparable analyses and produces a benefit-to-cost ratio of only 0.04 versus 0.15 for maternal vaccination.
8. Active surveillance infrastructure capable of detecting rare outcomes such as chorioamnionitis, stillbirth, and preterm delivery at the population level should be strengthened, given the structural limitations of passive systems like VAERS.
9. Conclusions
Maternal Tdap vaccination during the third trimester of pregnancy is superior to cocooning as an infant pertussis protection strategy on the grounds of relative effectiveness, absolute risk reduction, NNV, logistical tractability, and cost-effectiveness. Published cost-effectiveness analyses place maternal vaccination at $7,601 to $414,523 per QALY depending on model assumptions and epidemiological context, compared with $1,172,825 per QALY for cocooning in comparable United States analyses.
A genuinely objective appraisal must acknowledge that the gap between impressive relative effectiveness figures (78–93%) and modest absolute risk reductions (ARR 0.03–0.05% for cases and hospitalization; ARR ~0.00046% for death) reflects genuine epidemiological reality in contemporary high-coverage settings. The corresponding cost estimates — $14 million to $22 million per infant death prevented for maternal vaccination, and $65 million to $100 million for cocooning — should be presented transparently rather than obscured behind relative percentage figures.
The cocooning strategy should not be abandoned but should be understood as a supplementary measure in unusual circumstances, one whose very high NNV for infant death (>1,000,000) and consistent implementation failures make it insufficient as a standalone approach.
Future research priorities should include: adequately powered prospective studies of maternal Tdap safety with respect to the chorioamnionitis and postpartum haemorrhage signals; next-generation pertussis vaccines with greater and more durable protection; and routine reporting of absolute alongside relative effectiveness figures and cost-per-outcome estimates in all vaccine effectiveness studies.
Conflicts of Interest
The author declares no conflicts of interest.
Funding
This review received no specific funding from any public, commercial, or not-for-profit funding agency.
References
1. Vygen-Bonnet S, et al. Acellular pertussis vaccine components: Today and tomorrow. Pathogens. 2020;9(7):583. PMC7349526.
2. World Health Organization. Pertussis vaccines: WHO position paper. Wkly Epidemiol Rec. 2015;90(35):433-458.
3. Centers for Disease Control and Prevention. Manual for the Surveillance of Vaccine-Preventable Diseases. Chapter 10: Pertussis. Updated February 2026. https://www.cdc.gov/surv-manual/php/table-of-contents/chapter-10-pertussis.html
4. Gilley N, Goldman RD. Protecting infants from pertussis. Can Fam Physician. 2014;60(1):47-49. PMC3922557.
5. Malenka DJ, Baron JA, Johansen S, Wahrenberger JW, Ross JM. The framing effect of relative and absolute risk. J Gen Intern Med. 1993;8(10):543-548.
6. Skowronski DM, Janjua NZ, Tsafack EP, Ouakki M, Hoang L, De Serres G. The number needed to vaccinate to prevent infant pertussis hospitalization and death through parent cocoon immunization. Clin Infect Dis. 2012;54(3):318-327.
7. GBD 2013 Mortality and Causes of Death Collaborators. Global, regional, and national age-sex specific all-cause and cause-specific mortality for 240 causes of death, 1990-2013. Lancet. 2015;385(9963):117-171.
8. Acosta AM, et al. Lessons from a mature acellular pertussis vaccination program. Vaccines. 2022;10(3):435. PMC8989716.
9. Lam C, et al. Vaccine-induced selection pressure and pertactin-deficient Bordetella pertussis strains. Emerg Infect Dis. 2014;20(8):1248-1255.
10. Healy CM, Baker CJ. Prospects for prevention of childhood infections by maternal immunization. Curr Opin Infect Dis. 2006;19(3):271-276.
11. Advisory Committee on Immunization Practices. Prevention of pertussis, tetanus, and diphtheria with vaccines in the United States: Recommendations of the ACIP. MMWR Recomm Rep. 2018;67(RR-2):1-44.
12. Skowronski DM, Janjua NZ, Tsafack EP, Ouakki M, Hoang L, De Serres G. The number needed to vaccinate to prevent infant pertussis hospitalization and death through parent cocoon immunization. Clin Infect Dis. 2012;54(3):318-327.
13. Lugner AK, van der Maas N, van Boven M, Mooi FR, de Melker HE. Cost-effectiveness of targeted vaccination to protect newborns against pertussis: comparing neonatal, maternal, and cocooning vaccination strategies. Vaccine. 2013;31(46):5392-5397.
14. Urwyler P, Heininger U. Protecting newborns from pertussis: the challenge of complete cocooning. BMC Infect Dis. 2014;14:397.
15. DeSilva M, et al. Maternal Tdap vaccination: coverage and acute safety outcomes in the Vaccine Safety Datalink, 2007-2013. Vaccine. 2017;35(36):4761-4769. PMC6506839.
16. Eberhardt CS, et al. Maternal immunization earlier in pregnancy maximizes antibody transfer and expected infant seropositivity against pertussis. Clin Infect Dis. 2016;62(7):829-836.
17. Centers for Disease Control and Prevention. Vaccinating Pregnant Patients. Updated December 2025. https://www.cdc.gov/pertussis/hcp/vaccine-recommendations/vaccinating-pregnant-patients.html
18. Munoz FM, Bond NH, Maccato M, et al. Safety and immunogenicity of tetanus diphtheria and acellular pertussis (Tdap) immunization during pregnancy in mothers and infants: a randomized clinical trial. JAMA. 2014;311(17):1760-1769. PMC4333147.
19. Skoff TH, Blain AE, Watt J, et al. Impact of the US maternal Tdap vaccination program on preventing pertussis in infants <2 months of age. Clin Infect Dis. 2017;65(12):1977-1983.
20. American College of Obstetricians and Gynecologists. Update on immunization and pregnancy: tetanus, diphtheria, and pertussis vaccination. Committee Opinion No. 718. Obstet Gynecol. 2017;130:e153-e157.
21. Baxter R, Bartlett J, Fireman B, Lewis E, Klein NP. Effectiveness of vaccination during pregnancy to prevent infant pertussis. Pediatrics. 2017;139(5):e20164091.
22. Public Health Agency of Canada. Update on immunization in pregnancy with Tdap vaccine. https://www.canada.ca/en/public-health/services/publications/healthy-living/update-immunization-pregnancy-tdap-vaccine.html
23. Layton JB, Butler AM, Li D, et al. Prenatal Tdap immunization and risk of maternal and newborn adverse events. Vaccine. 2017;35(33):4072-4078. PMC5546155.
24. Chen C, et al. Risk signal assessment of Tdap vaccine use alone by pregnant women: an updated pharmacoepidemiological study. Front Cell Infect Microbiol. 2025;15:1689560. PMC12886459.
25. Sukumaran L, McCarthy NL, Kharbanda EO, et al. Association of Tdap vaccination with acute events and adverse birth outcomes among pregnant women with prior tetanus-containing immunizations. JAMA. 2015;314(15):1581-1587. PMC6586223.
26. Terranella A, Asay G, Messonnier M, Clark T, Liang J. Preventing infant pertussis: a decision analysis comparing prenatal vaccination to cocooning. Presented at 49th IDSA Annual Meeting. 2011. Boston, MA.
27. Hersh AR, Ameel AA, Packer CH, Caughey AB. Universal tetanus-diphtheria-pertussis vaccination during pregnancy: a cost-effectiveness analysis. Obstet Gynecol. 2023;141(3):547-556.
28. Ameel AA, Hersh AR, Packer CH, Caughey AB. 1053: Cost-effectiveness of Tdap vaccination during pregnancy. Am J Obstet Gynecol. 2020;222(1):S671-S672.
29. Atkins KE, Fitzpatrick MC, Galvani AP, Townsend JP. Cost-effectiveness of pertussis vaccination during pregnancy in the United States. Am J Epidemiol. 2016;183(12):1159-1170. PMC4908210.
30. Westra TA, de Vries R, Tamminga JJ, Sauboin CJ, Postma MJ. Cost-effectiveness analysis of various pertussis vaccination strategies primarily aimed at protecting infants in the Netherlands. Clin Ther. 2010;32(8):1479-1495.
31. Brophy J, Baclic O, Tunis MC, and: Pertussis vaccination in pregnancy in Canada: a cost-utility analysis. CMAJ Open. 2020;8(4):E651-E659.
32. Fernandez-Cano MI, Armadans Gil L, Campins Marti M. Cost-benefit of the introduction of new strategies for vaccination against pertussis in Spain. Vaccine. 2015;33(19):2213-2220.
33. Fernandes EG, Rodrigues CCM, Sartori AMC, De Soarez PC, Novaes HMD. Economic evaluation of adolescents and adults’ pertussis vaccination: a systematic review. Hum Vaccin Immunother. 2019;15(1):14-27. PMC6363086.
34. Terranella A, Asay GR, Messonnier ML, Clark TA, Liang JL. Pregnancy dose Tdap and postpartum cocooning to prevent infant pertussis: a decision analysis. Pediatrics. 2013;131(6):e1748-e1756.
35. Terranella A, Asay GR, Messonnier ML, Clark TA, Liang JL. Pregnancy dose Tdap and postpartum cocooning to prevent infant pertussis: a decision analysis. Pediatrics. 2013;131(6):e1748-e1756.