What They Don't Tell You About the Shingles Vaccine
The headline number is 97% effective. The number that actually matters to you is closer to 3%. Both are true. Here's what's going on — and why the distinction matters.
THE DISEASE
First, shingles is genuinely unpleasant
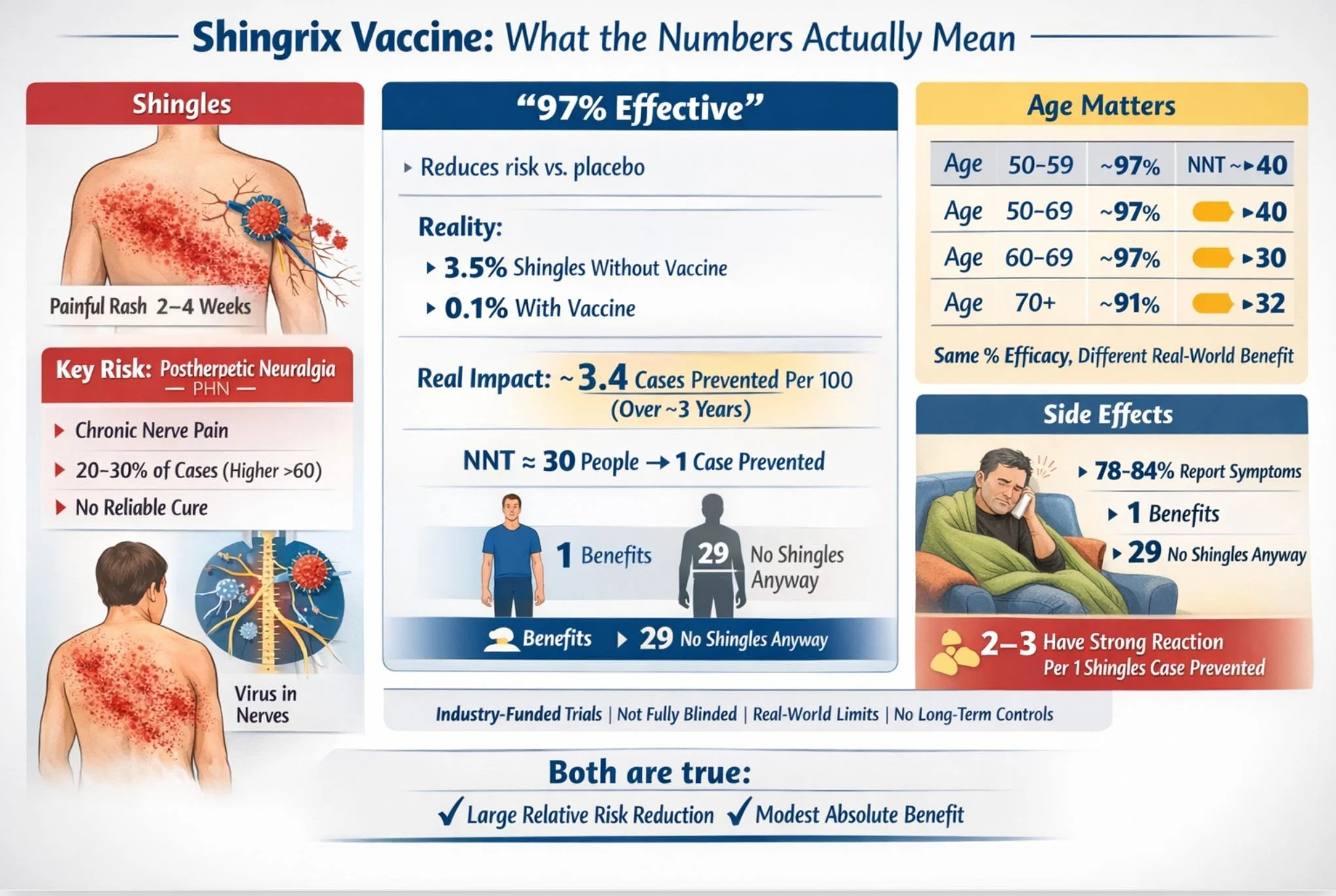
To fairly evaluate any vaccine, you have to start with the disease it prevents. Shingles is caused by the same virus that gave you chickenpox as a child, it never fully leaves your body. It retreats into your nervous system and can reactivate decades later, typically producing a painful, blistering rash that appears in a band on one side of your torso, face, or limbs.
For most people, the rash resolves in two to four weeks. But for a significant minority, roughly one in five people who get shingles, rising to nearly one in three among adults over 60, the pain doesn’t stop when the rash heals. This complication is called postherpetic neuralgia, or PHN. It’s a chronic nerve pain that can persist for months, years, or sometimes permanently, and is described by those who have it as burning, electric, and constant. It is associated with depression, sleep disruption, and social withdrawal.
Most cases of shingles are mild to moderate and resolve within a few weeks, with 10–20% becoming “severe” in a meaningful clinical sense.
I have personally had shingles fifteen plus years ago, an unusual and rather nasty variant called intra-abdominal shingles. Inside your abdomen. I did not have medical insurance, and so I delayed treatment until I could not stand the pain. As a consequence, I developed a particularly nasty postherpetic neuralgia- a paralytic polio-like syndrome that caused a partial paralysis of the left side of my abdomen. Suffice to say, this created problems as an advanced equestrian. I quite literally lost my core. That said, I personally consider that a “natural” booster, and have never accepted the shingles vaccine. My body, my choice.
About 10–18% of shingles cases progress to PHN, which is the main long-term issue. There is no “cure” for PHN, as no drug will cure it. The treatments available are nerve-calming drugs, like gabapentin, antidepressants used as pain medications, and lidocaine patches, which help some people, but roughly 40 to 50 percent of PHN patients never achieve adequate pain relief from any available therapy. BTW - stating that there is “no cure” is medically acceptable language, but it lacks nuance. Most people do improve and even achieve complete recovery, gradually over time. But there is no treatment that will “cure” PHN - drugs will only help with the symptoms, so this is a significant nuance.
This matters for understanding the vaccine because preventing shingles also means preventing any chance of developing PHN. But how effective is the vaccine at actually doing that for any individual?
THE NUMBERS
97% effective. What does that actually mean?
The figure comes from two large clinical trials called ZOE-50 and ZOE-70, which together enrolled about 30,000 adults across 18 countries. Participants were randomly assigned to receive either Shingrix or a saline injection, then followed for roughly three to four years to see who developed shingles.
Here are the actual results for adults aged 60 to 69, the age group with the highest shingles rates:
Source: ZOE-50 trial (Lal et al., N Engl J Med, 2015). Modified vaccinated cohort, mean follow-up 3.2 years.
Now you can see both numbers at once. The vaccine group’s rate was about 3% of the placebo group’s rate; that’s where “97% effective” comes from. It’s the ratio. It’s a relative measure.
But look at the placebo column. Only 3.5 out of 100 unvaccinated people in this age group developed shingles over roughly three years. That means 96.5 out of 100 would not have gotten shingles anyway, with or without the vaccine.
“97% effective” means the vaccine reduced the rate among those who would have gotten shingles. It does not mean 97 in 100 vaccinated people are protected from something that would otherwise have happened to them.
The absolute benefit, the actual reduction in cases attributable to vaccination, was about 3.4 cases prevented per 100 people vaccinated over three years. That translates to a “number needed to treat” (NNT) of approximately 30 people you need to vaccinate in this age group to prevent one case of shingles over a three-year period.
HOW TO READ NNT
If the NNT is 30, it means that for every 30 people vaccinated, 29 received no direct benefit from the vaccine over the trial period (because they wouldn’t have developed shingles anyway), and 1 was protected from a shingles case. The other 29 still took on whatever risks or side effects the vaccine entailed. NNT doesn’t tell you which person you’ll be; it tells you the odds.
How NNT varies by age
One of the genuinely useful features of Shingrix is that its relative efficacy doesn’t fall off with age the way the older vaccine did. The percentage reduction in risk is roughly similar whether you’re 55 or 80.
But, and this is important, the absolute benefit varies by age because older people face a higher baseline risk of shingles. More cases to prevent means more absolute benefit per vaccination
Relative efficacy from primary trial publications. Absolute reduction calculated from published incidence rates x trial follow-up duration. Real-world NNTs are likely somewhat higher than these trial-population figures.
Notice: the relative efficacy is almost identical across all ages. But the absolute numbers diverge because the baseline disease rate varies. A 97% reduction in a low-risk group gives you less absolute benefit than a 91% reduction in a high-risk group.
This is not a trick or a critique of the vaccine; it’s just how mathematics works. But it does mean that when you read “97% effective,” you should immediately ask: 97% of what? Of whom? Over how long?
THE SIDE EFFECTS
The side effects from the vaccine are real, and the trials likely undercounted them
Shingrix has a strong adjuvant, a chemical booster that makes your immune system respond more intensely. This is how it achieves such high efficacy even in older adults. The tradeoff is a higher rate of side effects than most vaccines.
In the trials, 78 to 84 percent of vaccinated people reported at least one reaction within 7 days, compared with about 30 percent in the placebo group. Most were mild: sore arm, tiredness, headache. But a meaningful proportion were severe enough to disrupt daily life. This is measured as “grade 3” reactions — fever over 39°C, muscle pain that prevents normal activity, and injection-site pain at rest.
The “number needed to harm” (NNH) for a grade 3 reaction, meaning one serious enough to stop you doing normal things for a day or two, was:
• Ages 50–69: approximately 1 in 11 vaccinated people
• Ages 70+: approximately 1 in 25 vaccinated people
Older adults have fewer severe reactions, not because the vaccine is gentler, but because their immune systems respond less intensely to stimulation. This is, slightly counterintuitively, the same reason the relative efficacy is a few percentage points lower in the oldest group.
Here’s the comparison that actually matters: across the three-year trial window, for every case of shingles prevented in the 60–69 age group, approximately 2.7 people experienced a grade 3 reaction. Those reactions lasted one to two days. Shingles and its complications can last far longer.
However, there is an important asterisk on all the side effect data: the trials were not fully blinded.
THE BLINDING PROBLEM
In a fully “double-blind” trial, neither the participant nor anyone treating them knows whether they received the real drug or the placebo. Shingrix couldn’t achieve this because the vaccine, when mixed, looks visibly different from clear saline. The nurse preparing the syringe knew what they were drawing up.
More importantly, the vaccine causes such distinctive reactions, a cluster of fever, fatigue, and muscle aches within 24 hours, that many participants who received it almost certainly figured out they were in the vaccine group. Research consistently shows that people who know (or strongly suspect) they got the active treatment tend to report more symptoms on diary cards. This means the side-effect numbers from the trial are probably somewhat higher than they would be in a perfectly blinded study. No one knows by how much.
THE EVIDENCE TRAIL
Every single trial was paid for by the company selling the vaccine
This is worth saying plainly, without drama: all of the clinical evidence for Shingrix, every pivotal trial, every extension study, every long-term follow-up analysis, was funded by GlaxoSmithKline, the vaccine’s manufacturer. Several named authors on the key publications were GSK employees at the time. Medical writing for the main publications was paid for by GSK. The decision about when to submit results for publication was made by GSK.
None of this is unusual. This is standard practice in the pharmaceutical industry, and it’s the only realistic way to fund trials of this size. But it does mean you’re relying on a company with a direct financial interest in a positive result for every piece of evidence about the product’s efficacy and safety. No independent, publicly-funded pivotal trial of Shingrix exists.
Industry funding doesn’t automatically mean the results are wrong. The trials used randomization, independent adjudication panels, and safety monitoring committees. Post-marketing studies conducted without GSK involvement have found broadly similar effectiveness figures. But those corroborating studies have their own limitations, and “broadly similar” still means lower effectiveness than the headline trial numbers. Real-world effectiveness tends to be 10 to 15 percentage points lower than controlled-trial efficacy because real patients are older, sicker, and on more medications than the trial cohort.
The long-term data has a significant flaw
GSK extended the trials with a follow-up study called ZOE-LTFU, which tracked participants for up to 11 years post-vaccination. The results looked good: roughly 88% relative efficacy maintained through year 11.
The problem: by the time the extension started, the original placebo group had already been offered the vaccine (because the main trial was over). There were no unvaccinated controls left. So the researchers had to calculate efficacy by comparing vaccinated participants against a projected placebo group based on earlier data, essentially asking: how many cases would we have expected if these people had never been vaccinated, using the old placebo rates?
This approach has real limitations. It assumes shingles rates didn’t change over time. It doesn’t account for the COVID-19 pandemic (which affected immune system dynamics and social mixing). It assumes the people who stayed in the study for 11 years are representative of everyone who started. None of these assumptions can be verified. GSK itself acknowledged in the publication that this methodology “could overestimate efficacy.”
IF YOU DO GET SHINGLES
The treatments for shingles work, if you get them quickly
If you do develop shingles, three antiviral medications: valacyclovir, famciclovir, and acyclovir, can reduce its severity and duration if you start them within 72 hours of the rash appearing. That window matters a great deal. After 72 hours, the benefit drops off significantly.
These antivirals reduce the severity of the shingles episode but don’t reliably prevent PHN. Once that nerve pain develops, treatment becomes much harder.
IF YOU DEVELOP PHN
The first-line treatments are gabapentin and pregabalin (anticonvulsants used as nerve pain drugs), tricyclic antidepressants like nortriptyline, and lidocaine patches applied to the skin. These help some people meaningfully. But approximately 40 to 50 percent of PHN patients don’t get adequate relief from any of them. Opioids are sometimes used but carry dependence risks that are especially significant in older adults. There is no cure.
THE BOTTOM LINE
So, should you get it?
This content is for paid subscribers only:
This is a genuine decision that depends on your age, your health, your risk tolerance, and how much weight you put on preventing a painful disease versus experiencing a day or two of vaccine-related misery. In other words, there should be true “shared decision making” and true “informed consent” when patients are offered a shingles vaccine. Not just posterboard advertisements in the Walmart pharmacy line. Here is what the data actually show:
The case for vaccination:
Shingles is genuinely unpleasant, and PHN is a serious complication that can become permanent and doesn’t respond reliably to treatment. Vaccination substantially reduces your relative risk of both. The absolute benefit is modest over any three-year window but grows meaningfully if the protection lasts a decade or longer, as the imperfect long-term data tentatively suggests. Serious, lasting harm from the vaccine was not detected in the trials, and the short-term reactions, though real, resolve in a day or two.
The case for skepticism:
Every piece of primary evidence comes from the manufacturer. The trials were not truly double-blind. The grade 3 reaction rate, roughly 1 in 11 for adults under 70, is high for a preventive intervention in generally healthy adults, and may be somewhat inflated by the blinding limitations. The NNT over a three-year window is 30 to 40, meaning most vaccinated people derive no personal benefit in that period. Long-term efficacy data come from an unblinded study without a control group.
What is not in dispute:
The 91 to 97 percent figure is real, but it is a relative measure. Relative risk reductions, especially large-sounding ones, are routinely used in pharmaceutical promotion because they sound more impressive than the absolute numbers. The absolute benefit: 2.5 to 3.4 cases prevented per 100 people vaccinated over three years is also real, and is the number that pertains to your individual decision.
A reasonable, well-informed person could go either way on this. What they shouldn’t do is make the decision based solely on “97% effective” without knowing what that number means, or doesn’t mean.
References
Source material: This summary is drawn from a narrative review of the ZOE-50 and ZOE-70 phase 3 trials, the ZOE-LTFU long-term follow-up study, independent systematic reviews (including the Therapeutics Initiative Letter 114), and published treatment guidelines. See below for the full analysis.
.Conflict of interest: none. The author has no financial relationship with GlaxoSmithKline or any competing vaccine manufacturer.
This is not medical advice. Individual decisions about vaccination should be made with a qualified clinician who knows your health history.
Efficacy, Safety, Benefit-Harm Analysis, and Pharmacological Treatment of Herpes Zoster: A Narrative Review Focused on the Recombinant Zoster Vaccine (Shingrix)
Running title: Shingrix Efficacy and Safety Review
Robert W. Malone MD, MS, with the analytical assistance of CLAUD
Conflict of Interest: None declared
Abstract
Background: Herpes zoster (shingles) and its principal complication, postherpetic neuralgia (PHN), impose substantial morbidity on older adults. The recombinant zoster vaccine (RZV; Shingrix, GlaxoSmithKline) received United States Food and Drug Administration approval in 2017 and was subsequently designated the preferred agent for shingles prevention by United States advisory bodies. This narrative review critically examines evidence from the pivotal ZOE-50 and ZOE-70 phase 3 trials and their long-term follow-up, with particular attention to age-stratified number needed to treat (NNT) and number needed to harm (NNH) metrics, conflicts of interest in the sponsoring body, blinding limitations, and the pharmacological treatment landscape for acute herpes zoster and PHN.
Methods: Published data from ZOE-50, ZOE-70, ZOE-LTFU, independent systematic reviews, post-marketing surveillance, and treatment guidelines for herpes zoster and PHN were synthesized. Absolute risk reduction (ARR) was used to calculate NNT and NNH values by age group. Conflict-of-interest disclosures were extracted from primary publications and extension studies.
Results: Over a median follow-up of 3.2 to 3.7 years, RZV reduced the relative risk of shingles by 91 to 97 percent across all age groups (vaccine efficacy, a relative measure), with no statistically significant decline across age groups in the trial populations studied.
These relative figures, widely cited in promotional and guideline materials, translate to absolute risk reductions of approximately 2.5 to 3.4 percent over the trial period, depending on age group, reflecting differences in baseline incidence rather than differences in vaccine mechanism.
Trial-derived NNTs ranged from approximately 30 (ages 60 to 69) to 40 (ages 50 to 59). Lifetime modeled NNTs, derived from manufacturer-associated cost-effectiveness analyses, ranged from 6 to 17. The NNH for grade 3 systemic reactions preventing normal activity was approximately 11 in adults aged 50 to 69 and approximately 25 in those aged 70 and older; the observer-blind design may have inflated these figures. Serious adverse events occurred at rates statistically indistinguishable from placebo. All pivotal trials were funded by the manufacturer, several named authors held GSK employment or equity interests, and the long-term extension study used an open-label design with historical controls rather than a concurrent placebo arm. For acute herpes zoster, three antiviral agents are effective when initiated within 72 hours of rash onset; benefit beyond this window is uncertain. PHN treatment is of limited and variable efficacy, with approximately 40 to 50 percent of patients failing to achieve adequate relief from available agents.
Conclusions: Within the constraints of exclusively manufacturer-funded trials, RZV was measured to reduce the relative risk of shingles by 91 to 97 percent across studied age groups over a 3.5-year trial horizon; in absolute terms, this corresponded to approximately 2.5 to 3.4 cases prevented per 100 vaccinees over that period, with some attenuation of the relative efficacy evident in the open-label long-term follow-up. The trials were observer-blind rather than double-blind: injection preparers were unblinded, and participant inference of treatment assignment from the distinctive reactogenicity of the AS01B adjuvant was highly probable, potentially inflating solicited adverse event rates. These are not trivial limitations; they affect the certainty with which both efficacy and safety estimates can be accepted. The treatment landscape for PHN, in which no curative therapy exists and a substantial proportion of patients fail all available agents, is relevant context for individual benefit-harm decision-making but does not itself validate the trial evidence. The overall benefit-harm profile appears favorable in the reviewed data, with the important caveat that all primary evidence derives from a single commercial sponsor.
Keywords: herpes zoster; shingles vaccine; Shingrix; recombinant zoster vaccine; number needed to treat; number needed to harm; postherpetic neuralgia; antiviral therapy; gabapentin; pregabalin; conflict of interest; ZOE-50; ZOE-70
Introduction
Herpes zoster, commonly known as shingles, results from the reactivation of latent varicella-zoster virus (VZV) in the dorsal root or cranial nerve ganglia, typically decades after primary varicella infection.1 The lifetime risk of herpes zoster in the United States approaches one in three individuals, with the highest incidence occurring among adults aged 60 and older.2 The most common and debilitating complication is PHN, characterized by persistent neuropathic pain that may last months to years after rash resolution and that occurs in approximately 10 to 40 percent of shingles episodes.3
Prior to 2017, the only available vaccine was the live-attenuated zoster vaccine (Zostavax, Merck), which demonstrated a relative risk reduction of approximately 51 percent for shingles and 67 percent for PHN in adults aged 60 and older, with this relative efficacy declining sharply with advancing age.4 RZV is a non-live recombinant subunit vaccine containing VZV glycoprotein E and the AS01B adjuvant system. It was approved by the FDA in October 2017 for adults aged 50 and older, and regulatory agencies and advisory bodies in the United States subsequently designated it as the preferred agent for shingles prophylaxis in immunocompetent adults and in immunocompromised adults aged 18 and older.5
Vaccine efficacy, as conventionally reported in clinical trials and regulatory submissions, is a relative measure: it expresses the proportional reduction in disease risk in the vaccinated group compared with the placebo group. While relative measures of efficacy tend to appear more impressive than absolute measures, clinicians and patients require absolute risk reduction (ARR) and the derived number needed to treat (NNT) to engage in meaningful shared decision-making. This review presents both relative and absolute effect estimates from the pivotal trials, examines the divergence between them across age groups, evaluates the evidence for long-term durability, critically appraises conflicts of interest and methodological limitations, and summarizes the pharmacological treatment options for acute herpes zoster and PHN.
Methods
A narrative review of published literature was conducted using PubMed, MEDLINE, and ClinicalTrials.gov. Search terms included “herpes zoster vaccine,” “Shingrix,” “recombinant zoster vaccine,” “ZOE-50,” “ZOE-70,” “number needed to treat,” “postherpetic neuralgia,” and “vaccine safety.” Primary data were drawn from the ZOE-506 and ZOE-707 phase 3 trial publications, the ZOE-LTFU extension study,8,9 and independent systematic reviews.10,11 Vaccine efficacy (VE) as reported in the primary publications is a relative measure, calculated as VE = 1 − (incidence in vaccine group / incidence in placebo group), and is equivalent to the relative risk reduction (RRR). Absolute risk reduction (ARR) was calculated independently from reported incidence rates as ARR = (placebo incidence rate − vaccine incidence rate) × mean follow-up duration, expressed as a percentage of vaccinees over the trial period. NNT was computed as 1/ARR. NNH values were computed as the reciprocal of the absolute risk increase for specific adverse events. Conflict-of-interest disclosures were extracted from all primary publications and extension study reports.
The ZOE Clinical Trial Program
ZOE-50: Adults Aged 50 and Older
ZOE-50 was a multinational, randomized, observer-blind, placebo-controlled phase 3 trial conducted at 167 centers across 18 countries.6 A total of 15,411 immunocompetent adults aged 50 and older were enrolled and randomized 1:1 to receive two intramuscular doses of RZV or saline placebo administered 2 months apart. Participants were stratified into three age groups (50 to 59, 60 to 69, and 70 years and older) with a mean follow-up of 3.2 years. The primary endpoint was confirmed herpes zoster incidence in the modified total vaccinated cohort.
The primary endpoint result was a reported vaccine efficacy of 97.2 percent (95% CI: 93.7 to 99.0; p < 0.001), a relative measure indicating that vaccinated participants developed confirmed herpes zoster at approximately 3 percent of the rate observed in the placebo group. Age-subgroup relative efficacy ranged from 96.6 percent in adults aged 50 to 59 to 97.9 percent in those aged 60 to 69.6 In absolute terms, the underlying incidence rates were 0.3 cases per 1000 person-years in the vaccine group versus 9.1 per 1000 person-years overall in the placebo group, translating to approximately 2.5 to 3.4 cases prevented per 100 vaccinees over the 3.2-year follow-up period depending on age subgroup (see Table 1). Relative efficacy was consistent across age subgroups with no statistically significant decline with advancing age, in contrast to Zostavax. Grade 3 adverse reactions were reported by 17.0 percent of vaccine recipients versus 3.2 percent of placebo recipients, a substantially higher rate that the observer-blind design may have inflated through differential symptom reporting.
ZOE-70: Adults Aged 70 and Older
ZOE-70 was conducted concurrently at the same sites with an identical protocol, enrolling 13,900 adults aged 70 and older (mean age 75.6 years, range 62 to 96 years) with a mean follow-up of 3.7 years.7 Reported relative vaccine efficacy was 89.8 percent (95% CI: 84.2 to 93.7; p < 0.001) in ZOE-70 alone, and 91.3 percent (95% CI: 86.8 to 94.5) in a prespecified pooled analysis of adults aged 70 and older from both trials. In absolute terms, herpes zoster occurred in 23 vaccine recipients and 223 placebo recipients (0.9 versus 9.2 per 1000 person-years), translating to approximately 3.1 cases prevented per 100 vaccinees over the 3.7-year follow-up period.7 Relative efficacy was similar in adults aged 70 to 79 (90.0%) and those aged 80 and older (89.1%), with no statistically significant age-dependent decline within this older cohort. Relative vaccine efficacy against PHN was 88.8 percent (95% CI: 68.7 to 97.1) in the pooled analysis; the wide confidence interval reflects the small number of PHN events even in this large trial, and no absolute PHN incidence reduction can be robustly estimated from so few events.
One methodological issue of note: 865 of 14,816 enrolled ZOE-70 participants were excluded from all analyses because of deviations from Good Clinical Practice standards at one study site, which was subsequently closed. One additional site was closed for financial reasons.7 These post-randomization exclusions represent approximately 6 percent of the enrolled population, and while the exclusion of compromised data is appropriate, the scale of exclusions warrants acknowledgment as a potential source of selection bias.
Long-Term Follow-Up (ZOE-LTFU)
Former ZOE-50 and ZOE-70 vaccinees who received at least one RZV dose were offered enrollment in an open-label phase 3b extension study (ZOE-LTFU) beginning approximately 5 years post-vaccination. The final analysis, published in 2025, evaluated 7,273 participants through 11 years post-vaccination.9 Reported relative vaccine efficacy against herpes zoster was 87.7 percent (95% CI: 84.9 to 90.1) cumulative from dose 2 through the end of ZOE-LTFU in participants aged 50 and older, with a point estimate of 82.0 percent (95% CI: 63.0 to 92.2) in the eleventh year post-vaccination. These figures are relative risk reductions calculated against a historical control rather than a concurrent placebo arm, which substantially limits their interpretability (see below). Reported relative efficacy against PHN was 87.5 percent. No vaccine-related serious adverse events were attributed to RZV during ZOE-LTFU, though the unblinded, uncontrolled design substantially limits the strength of any safety inference from this phase of the program.
Important limitations of ZOE-LTFU bear explicit statement. The extension study was open-label, unblinded, and lacked a concurrent placebo arm. Efficacy was calculated against a historical control constructed from ZOE-50 and ZOE-70 placebo-group data, which introduces several assumptions: that baseline shingles incidence in the placebo population would have remained stable, that no secular trends in herpes zoster incidence occurred, and that enrollment into ZOE-LTFU was not systematically biased toward healthier or more compliant participants. Additionally, herpes zoster cases occurring during the gap period between ZOE-50/70 and ZOE-LTFU enrollment were not captured in the primary efficacy analysis.9
Age-Stratified Efficacy: Number Needed to Treat
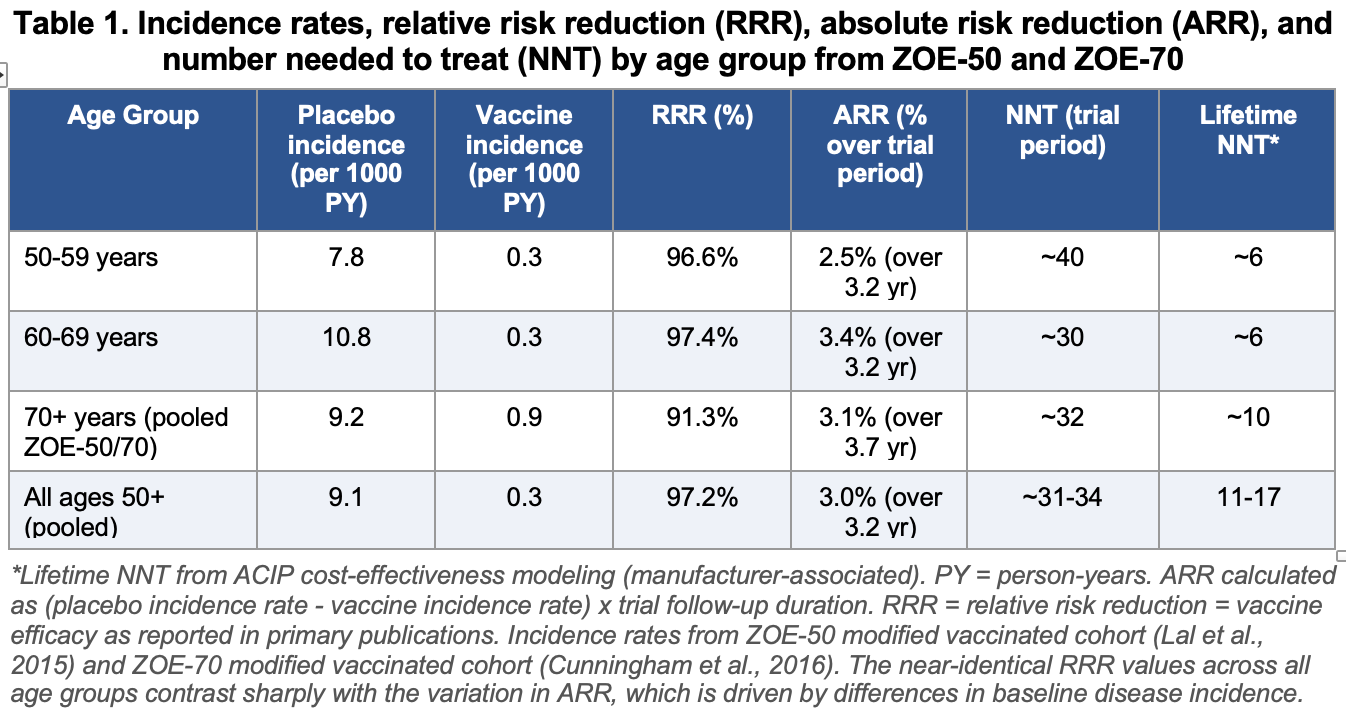
Table 1 presents both the relative vaccine efficacy (RRR, as reported in the primary publications) and the absolute risk reduction (ARR, calculated from the raw incidence rates) by age group. Presenting both measures together allows the divergence between them to be directly appraised rather than obscured by selective reporting of the more impressive relative figures. All vaccine efficacy figures cited in the primary publications are relative measures.
Table 1 presents the primary efficacy data in a form that permits direct comparison of relative and absolute measures. This comparison is essential because the two metrics can create substantially different impressions of the same data, and understanding that divergence is a prerequisite for meaningful clinical interpretation.
Relative Risk Reduction: What It Shows and What It Conceals
The RRR, synonymous with “vaccine efficacy” as conventionally reported in trial publications and regulatory documents, is the proportional reduction in disease risk in the vaccinated group relative to the placebo group. It is calculated as 1 minus the ratio of incidence rates and answers the question: by what fraction was the rate of disease reduced? Across all three age groups in ZOE-50 and ZOE-70, RZV produced RRRs in the range of 91 to 97 percent, with no statistically significant difference between age groups.6,7 This consistency in the relative measure is prominently cited in promotional and guideline materials as a key differentiator from Zostavax, whose RRR declined markedly with age. It is important to recognize, however, that a consistent RRR across age groups does not imply a consistent absolute benefit: when baseline risk differs between groups, the same RRR produces very different absolute reductions.
The clinical limitation of the RRR is that it contains no information about baseline risk. A 97 percent RRR means something very different to a 50-year-old with an annual shingles incidence of 3 per 1000 than it does to an 80-year-old with an annual incidence of 12 per 1000. In the first case, the vaccine prevents approximately 2.9 cases per 1000 person-years; in the second, approximately 11.6 cases per 1000 person-years. The RRR is the same; the absolute benefit differs fourfold. Reporting RRR alone, or leading with it as the primary efficacy metric, systematically overstates the practical value of the intervention to lower-risk individuals and obscures the variation in absolute benefit that is directly relevant to clinical decision-making and policy.
Absolute Risk Reduction: Age-Stratified Results and Their Interpretation
The ARR is calculated from the incidence data reported in Table 1 as the difference between the placebo and vaccine group event rates multiplied by the trial follow-up duration, yielding cumulative incidence prevented per 100 vaccinees over that period. The ARR varies substantially by age group, driven entirely by differences in baseline shingles incidence rather than any difference in vaccine mechanism:
In the 50 to 59 age group (ZOE-50), the placebo incidence was 7.8 per 1000 person-years and the vaccine incidence was 0.3 per 1000 person-years, a difference of 7.5 per 1000 person-years. Over the 3.2-year mean follow-up, this translates to approximately 2.5 prevented cases per 100 vaccinees, or an NNT of approximately 40.6
In the 60 to 69 age group (ZOE-50), the placebo incidence was 10.8 per 1000 person-years versus 0.3 per 1000 person-years in the vaccine group, a difference of 10.5 per 1000 person-years. Over 3.2 years, this yields approximately 3.4 prevented cases per 100 vaccinees, NNT approximately 30. This is the most favorable absolute effect among the three groups, reflecting the peak population incidence in this age range.6
In adults aged 70 and older (pooled ZOE-50 and ZOE-70 data), the placebo incidence was 9.2 per 1000 person-years versus 0.9 per 1000 person-years in the vaccine group, a difference of 8.3 per 1000 person-years. Over the 3.7-year mean follow-up in ZOE-70, this yields approximately 3.1 prevented cases per 100 vaccinees, NNT approximately 32. The placebo incidence in this group is somewhat lower than in the 60 to 69 group in ZOE-50, which may reflect differences between the trial populations rather than a true biological reversal of the age-incidence relationship; population-based data consistently show shingles incidence rising steeply beyond age 70.7
The Divergence Between RRR and ARR: Clinical Implications
The contrast between the near-uniform RRR (91 to 97 percent across all ages) and the varying ARR (2.5 to 3.4 percent over the trial period, depending on age) illustrates a well-documented phenomenon in evidence-based medicine: high relative risk reductions can coexist with modest absolute risk reductions when baseline risk is low. This is not a criticism of the vaccine’s mechanism of action; it is a mathematical consequence of the relationship between relative and absolute measures that applies to any intervention in any disease with moderate baseline incidence.
From a communication standpoint, the practical meaning of these ARR figures is that over approximately 3.2 years, between 97 and 98 out of every 100 vaccinated adults in the trial populations would not have developed shingles regardless of vaccination, while 2 to 3 in 100 avoided shingles attributable to the vaccine. Framed this way, the intervention’s individual-level benefit is modest over a short horizon, even as its population-level impact across millions of eligible adults may be substantial. Both framings are arithmetically correct; they emphasize different aspects of the same data.
The appropriate time horizon further complicates interpretation. The trial-derived ARRs above reflect approximately 3 to 4 years of observation. If protection persists at high efficacy over a decade or more, as the open-label extension data tentatively suggest, then the cumulative ARR over a lifetime is substantially larger and the NNT correspondingly lower. However, as discussed in the long-term follow-up section, the absence of a concurrent placebo group in ZOE-LTFU means that long-horizon ARR estimates rest on historical controls and modeling assumptions rather than direct observation. Lifetime NNT estimates of 6 to 17 from ACIP modeling should be understood as projections with uncertainty, not as empirically measured effects.
It is also worth noting that the incidence figures in ZOE-50 and ZOE-70 are derived from populations who were specifically selected to be immunocompetent, free of prior shingles, and generally healthy. Population-based incidence data, and real-world effectiveness studies, consistently show lower observed effectiveness than trial efficacy estimates, suggesting that the ARRs presented here likely represent an upper bound on absolute benefit in clinical practice.
Age-Stratified Safety: Number Needed to Harm
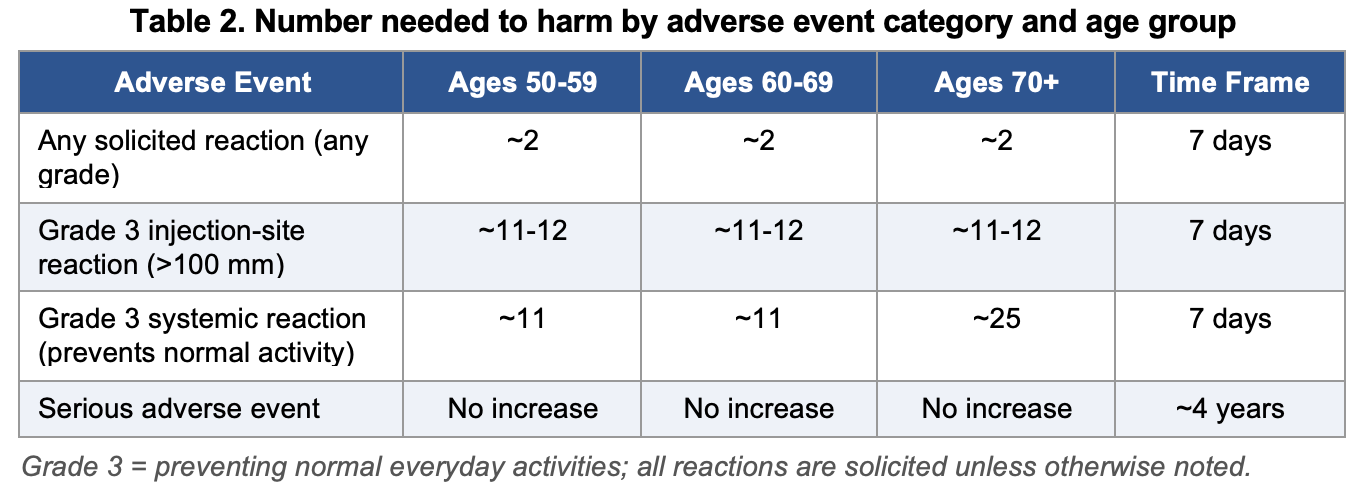
Table 2 summarizes the NNH for key adverse outcomes by age group, derived from ZOE-50, ZOE-70, and pooled safety analyses.6,7,11,13
Reactogenicity was the dominant safety signal. The AS01B adjuvant system elicits a vigorous innate immune response, and local reactions (injection-site pain, redness, and swelling) and systemic reactions (myalgia, fatigue, headache, shivering, and fever) were substantially more common in vaccine recipients than in placebo recipients within the 7-day post-vaccination window, with 78 to 84 percent of vaccinees reporting at least one solicited reaction compared with approximately 30 percent of placebo recipients.13 As noted in the blinding section, these rates must be interpreted with caution: the observer-blind design, with its highly reactive adjuvant and clear saline placebo, creates conditions in which differential symptom reporting by participants who infer their treatment assignment is likely. The NNH for grade 3 systemic reactions differs by age: approximately 11 in adults aged 50 to 69 and approximately 25 in those aged 70 and older.
The higher rate of severe reactions in younger adults reflects the more vigorous immune response to the AS01B adjuvant before the attenuating effects of immunosenescence take hold. The median duration of grade 3 reactions was 1 to 2 days. No evidence of prolonged or permanent harm attributable to these reactions was identified in the trial program, though the open-label design of ZOE-LTFU limits the strength of that safety inference in the long-term period.13
For serious adverse events, defined as events associated with hospitalization, disability, or life-threatening outcomes, no statistically significant difference was observed between vaccine and placebo recipients across any trial (relative risk 0.97; 95% CI: 0.91 to 1.05).13 This finding was consistent across ZOE-50 and ZOE-70. Rare events of special interest, including Guillain-Barre syndrome, were identified in post-marketing surveillance: a postmarketing observational study found an increased risk of Guillain-Barre syndrome in the 42 days following RZV vaccination. The absolute risk increment is small, but it is a genuine signal identified independently of the manufacturer-controlled trial program and warrants disclosure in clinical discussions.14
Integrated Benefit-Harm Analysis
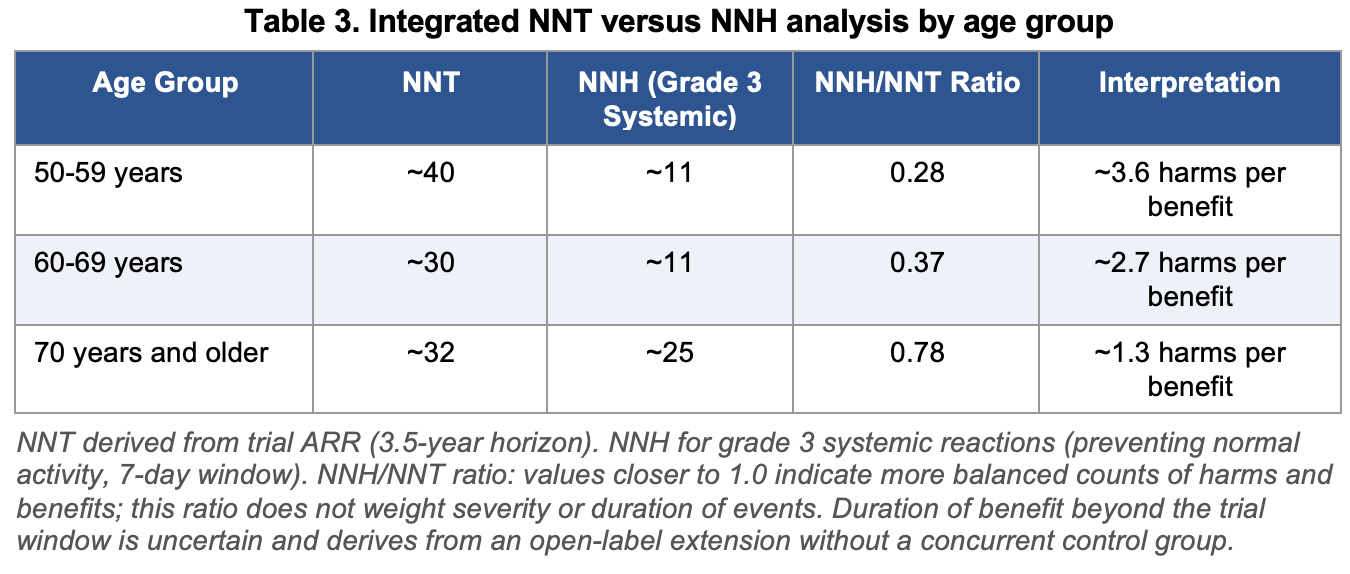
Table 3 presents an integrated comparison of NNT and NNH across age groups, using grade 3 systemic reactions (the most clinically meaningful transient harm) as the NNH reference event.
The NNH/NNT ratio conveys a useful but necessarily simplified picture. Over a 3.5-year trial horizon, for every shingles case prevented in adults aged 60 to 69, approximately two individuals will experience a grade 3 systemic reaction lasting 1 to 3 days, while in adults aged 70 and older the ratio of harms to prevented cases narrows to approximately 0.78. This raw comparison does not account for the asymmetry in event severity or duration: the grade 3 reactions are transient, while shingles and its potential complication of PHN can be prolonged and severe. However, the ratio also does not account for the fact that the NNH may be inflated by the observer-blind design, that the NNT will be higher in real-world populations than in the trial-selected cohort, and that most vaccinated individuals derive no direct personal benefit over the trial window. The lifetime NNT estimates of 6 to 17 present a more favorable picture but rest on modeling assumptions that cannot be independently verified from the published trial data.
Conflicts of Interest in the ZOE Trial Program
The evidence base for RZV efficacy and safety rests almost entirely on manufacturer-sponsored research, which warrants systematic appraisal.
Funding and Sponsor Involvement
Both ZOE-50 and ZOE-70 were funded exclusively by GlaxoSmithKline Biologicals.6,7 For ZOE-LTFU, GSK’s involvement extended to study design, data collection, data analysis, data interpretation, writing of the report, and the decision to submit for publication.9 This degree of sponsor engagement at every stage of the research pipeline represents the highest level of potential influence on outcome reporting.
Author Conflicts
Multiple named investigators across ZOE publications were GSK employees at the time of the research, holding GSK equity positions in addition to employment.7,9 Medical writing for ZOE-70 was provided by 4Clinics, France, with funding support from GlaxoSmithKline Biologicals.7 Industry-sponsored medical writing has been identified in the literature as a mechanism through which sponsor preferences can influence the framing, emphasis, and selective reporting of trial results.
Blinding: Observer-Blind Versus Double-Blind
A critical and underappreciated methodological issue concerns the nature of blinding in the ZOE trials. Both studies are described in their primary publications as “observer-blind” rather than double-blind,6,7 a distinction that carries important implications for the validity of the safety and reactogenicity data.
The placebo used in both trials was 0.9% saline solution.6,7 Shingrix, however, requires reconstitution at the time of administration: the lyophilized glycoprotein E antigen must be mixed with the liquid AS01B adjuvant immediately before injection, producing a cloudy, opalescent suspension that is visually distinct from clear saline. As a consequence, the personnel preparing the syringes necessarily knew which intervention they were handling. The trials managed this by having the preparer stand behind a barrier or screen so that neither the participant nor the assessing clinician could see the filled syringe at the point of administration. The preparers themselves, however, remained functionally unblinded throughout the trial.
This arrangement means the ZOE trials achieved blinding of participants, treating clinicians, and endpoint adjudicators, but not of the individuals who prepared and drew up each injection. This falls short of the double-blind standard, under which both participants and all personnel interacting with the intervention are blinded to treatment allocation. The Consolidated Standards of Reporting Trials (CONSORT) guidelines recommend that trial reports specify precisely who was blinded to what information, and how well each blind succeeded, rather than relying on terms such as “double-blind” that may obscure the actual structure of masking.15 The ZOE publications do not report any formal assessment of blinding integrity, such as asking participants or clinicians to guess their treatment assignment at study end, which the CONSORT guidelines recommend.
A second, practically unavoidable source of unblinding was participant inference. Because approximately 78 to 84 percent of vaccine recipients experienced solicited local or systemic reactions within seven days of each dose, compared with approximately 30 percent of saline recipients,6,7 many vaccinees would have been able to deduce their group assignment from the intensity of their own post-injection symptoms, particularly fever, myalgia, fatigue, and prominent injection-site pain occurring together within 24 hours of vaccination. This form of de facto unblinding is essentially inherent to any vaccine trial using a highly reactogenic adjuvant system and cannot be fully prevented by procedural measures alone.
The consequences of imperfect blinding are outcome-dependent. For the primary efficacy endpoint, confirmed herpes zoster adjudicated by an independent blinded committee using clinical criteria and PCR testing, the blinding limitations are relatively inconsequential: a participant who correctly infers that they received the vaccine does not thereby alter whether they develop a dermatomal vesicular rash. The efficacy results are therefore unlikely to be substantially biased by the observer-blind design.
For the solicited adverse event data, however, the implications are more significant. Participants who knew or strongly suspected they were in the vaccine arm may have been more vigilant in monitoring symptoms, more likely to attribute minor aches, fatigue, and local discomfort to the injection, and more likely to record them on diary cards. Research on blinding and outcome reporting has consistently demonstrated that participant awareness of treatment allocation can influence self-reported outcome data, particularly for subjective endpoints such as pain and fatigue.16 The NNH estimates for reactogenicity reported from the ZOE trials may therefore be somewhat inflated relative to what a fully double-blind design would have found. Neither ZOE-50 nor ZOE-70 reported a blinding integrity assessment, so the magnitude of this potential bias cannot be quantified from the published data. The Therapeutics Initiative independent systematic review flagged the questionable blinding as a limitation of both studies.10
It bears noting that truly double-blind vaccine trials using a visually matched placebo formulation are methodologically achievable when resources permit. Some trials have used adjuvant-only controls (omitting the antigen) or have employed centralized masked syringe preparation with opaque syringes and covers to preserve blinding at the point of administration. Neither approach was used in the ZOE program. The practical and cost implications of scaled blinding in 30,000-participant multinational trials are not trivial, but the tradeoff is that the solicited safety data carry a degree of uncertainty that is not fully captured in the published effect estimates.
Long-Term Follow-Up Design
The absence of a concurrent placebo arm in ZOE-LTFU is a fundamental limitation that the manufacturer itself acknowledged explicitly. The use of historical controls from ZOE-50 and ZOE-70 placebo groups is susceptible to several sources of bias: secular trends in herpes zoster incidence may differ from those projected using historical data; the COVID-19 pandemic and associated changes in social behavior and immune exposure occurred during the follow-up period and were not adjusted for; enrollment into ZOE-LTFU was voluntary and included only approximately 50 percent of eligible vaccinees, creating potential for selection of healthier participants; and HZ cases occurring in the gap between ZOE-50/70 and ZOE-LTFU were not included in the primary analysis, potentially inflating apparent efficacy estimates.9
Contextualizing Industry Sponsorship
It is worth noting that industry-sponsored trials are not automatically invalid. The ZOE program employed randomization, endpoint adjudication by blinded committees, independent data and safety monitoring boards, registration on ClinicalTrials.gov, and disclosure of protocol and statistical analysis plans. Post-marketing real-world effectiveness studies conducted independently of GSK have found effectiveness in broadly similar ranges to the trial efficacy estimates, lending some external support to the direction of the effect, though real-world studies also have their own methodological limitations including confounding by indication and incomplete ascertainment of outcomes.12 Nevertheless, the complete absence of publicly funded, independent pivotal trials and the pervasive involvement of GSK employees in data analysis and manuscript preparation mean that readers are relying on a single financially interested party for all primary evidence. This is not unusual in pharmaceutical research, but it is a genuine constraint on the certainty of conclusions that should not be minimized by appeals to post-marketing corroboration.
Pharmacological Treatment of Herpes Zoster and Postherpetic Neuralgia
The pharmacological management of herpes zoster and its complications falls into two distinct phases: treatment of the acute episode and management of PHN when it develops. Both phases present important limitations that bear directly on the individual benefit-harm calculus of prevention decisions.
Antiviral Therapy for Acute Herpes Zoster
Three oral antiviral agents are licensed and guideline-recommended for the treatment of acute herpes zoster in immunocompetent adults: acyclovir, valacyclovir, and famciclovir.17 All three act by inhibiting VZV DNA polymerase, thereby suppressing viral replication and limiting the extent of neuronal and cutaneous injury. When initiated within 72 hours of rash onset, antiviral therapy accelerates lesion healing, reduces the formation of new vesicles, decreases viral shedding, and diminishes the severity of acute pain.17,18 The 72-hour window is critical: data supporting antiviral benefit relate primarily to patients treated within this timeframe, and efficacy is substantially diminished with later initiation, though treatment beyond 72 hours may still be considered if new lesions are continuing to form or if involvement includes the eye, ear, or cranial nerves.
Valacyclovir (1,000 mg three times daily for 7 days) and famciclovir (500 mg three times daily for 7 days) are preferred over acyclovir (800 mg five times daily for 7 to 10 days) in most clinical settings due to their more convenient dosing regimens and improved bioavailability.18 Head-to-head comparisons have demonstrated therapeutic equivalence among these three agents for cutaneous healing and pain resolution in immunocompetent patients, so practical factors including cost, formulary availability, and renal function guide agent selection.18 In patients with renal impairment, dose adjustment is required for all three agents, and acyclovir carries a particular risk of crystalline nephropathy with intravenous formulations.
In Europe, brivudine (125 mg once daily for 7 days) is also licensed for herpes zoster treatment. Its once-daily dosing and high oral bioavailability (approximately 90 percent) are advantages, and some evidence suggests faster onset of pain relief in severe cases compared with valacyclovir.19 However, brivudine carries an absolute contraindication in patients receiving 5-fluorouracil or other fluoropyrimidine chemotherapy due to a potentially fatal pharmacodynamic interaction, and it is not available in the United States.19
Corticosteroids are sometimes used as adjuncts to antiviral therapy to reduce acute pain and accelerate healing, but their role is narrow. They do not reduce the incidence or severity of PHN, they should never be used without concurrent antiviral coverage, and they carry particular risks in older patients including hyperglycemia, fluid retention, and immunosuppression that may theoretically permit viral dissemination.17
The principal limitation of antiviral therapy is that, while it reduces the burden of acute disease, it does not reliably prevent PHN. Meta-analyses and clinical trials have demonstrated that antivirals reduce the duration of pain after the acute episode, but a substantial proportion of treated patients still develop persistent pain.18
The Burden of Postherpetic Neuralgia and the Limits of Treatment
PHN is defined as pain persisting in the affected dermatome for 90 days or more after the onset of the herpes zoster rash. It occurs in approximately 10 to 20 percent of all shingles episodes overall, rising to 20 to 40 percent in adults aged 60 and older, and represents one of the most debilitating and treatment-refractory chronic pain conditions encountered in clinical practice.3 The pathophysiology involves persistent central and peripheral sensitization following VZV-induced damage to sensory nerve fibers and the spinal dorsal horn, a process that antiviral treatment cannot fully interrupt once established.
PHN pain is characteristically severe, constant or lancinating, frequently accompanied by allodynia (pain elicited by normally innocuous stimuli such as light touch or clothing), and profoundly disruptive to sleep, mood, and daily function. Pain may persist for months to years, and in a subset of patients it is essentially permanent. The condition has well-documented associations with depression, social isolation, falls, and decreased quality of life, disproportionately in older adults who are already at elevated risk from these consequences.3
There is no cure for PHN. Approximately 40 to 50 percent of patients with PHN fail to achieve adequate pain relief from any currently available treatment, and many patients who do respond require multiple agents or experience intolerable side effects that limit dosing to subtherapeutic levels.20
Pharmacological Treatment of Postherpetic Neuralgia
Multiple guidelines from the American Academy of Neurology, the Neuropathic Pain Special Interest Group of the International Association for the Study of Pain, and the European Federation of Neurological Societies converge on a tiered treatment approach for PHN, with the caveat that evidence from head-to-head comparisons among agents is sparse and most pivotal trials were also industry-sponsored.21
First-line agents include the alpha-2-delta ligands gabapentin and pregabalin, tricyclic antidepressants (TCAs), and the topical lidocaine 5% patch.21 Gabapentin (titrated to 1,800 to 3,600 mg per day in divided doses) has an NNT of approximately 8 for 50 percent pain reduction compared with placebo. Pregabalin (300 to 600 mg per day), with a more linear pharmacokinetic profile than gabapentin, has an NNT of approximately 4 for the same endpoint and may achieve therapeutic effect more rapidly.22 Both agents require gradual dose titration over days to weeks to minimize somnolence, dizziness, and peripheral edema, which are particularly problematic in older adults. Gabapentin enacarbil, a prodrug of gabapentin with more predictable absorption, carries FDA approval specifically for PHN.
TCAs, including amitriptyline, nortriptyline, and desipramine, have established efficacy in PHN through modulation of descending noradrenergic and serotonergic pain-inhibitory pathways, as well as sodium channel blockade. Nortriptyline and desipramine are preferred over amitriptyline in older adults because they carry a lower burden of anticholinergic and cardiac adverse effects, though all TCAs require caution in patients with cardiovascular disease, urinary retention, glaucoma, or cognitive impairment.21
The topical lidocaine 5% patch provides localized analgesia through sodium channel blockade in sensitized peripheral nerve fibers, with minimal systemic absorption and therefore an advantageous tolerability profile in older or medically complex patients. It is particularly suited to PHN pain that is localized, of limited extent, and characterized by allodynia.21
Second- and third-line options include opioid analgesics, tramadol, and high-concentration capsaicin (the capsaicin 8% patch, Qutenza). Opioids provide analgesic benefit supported by randomized controlled trial evidence but carry well-recognized risks of dependence, sedation, falls, and respiratory depression that are amplified in older adults.21 The capsaicin 8% patch achieves pain relief through depletion of substance P from cutaneous nociceptors following an intense but transient burning sensation at application; it requires clinic-based administration under topical anesthesia and the analgesic benefit, while sometimes meaningful, is inconsistent across patients.21
Multimodal combination therapy is increasingly favored when monotherapy fails, as drugs acting through different mechanisms can achieve additive analgesia at lower individual doses, potentially reducing adverse effects. The combination of a gabapentinoid with a topical agent is particularly useful given the absence of pharmacokinetic interactions and the low systemic burden of topical treatments.21
The Gap Between Evidence and Practice
Real-world prescribing patterns for PHN deviate substantially from guideline recommendations. In large claims database analyses, opioids remained the most frequently prescribed initial treatment for PHN despite being classified as second- or third-line agents, while fewer than 40 percent of patients received a guideline-recommended first-line agent as initial therapy.23 This disconnect likely reflects multiple factors: unfamiliarity with the PHN-specific evidence base, reluctance to prescribe TCAs in older adults due to cardiac and anticholinergic concerns, the slow titration requirements of gabapentinoids, and the relative ease of opioid prescribing in patients already presenting with severe pain.
The treatment data described above are relevant context for individual benefit-harm deliberation regarding vaccination, but they require careful framing. Comparisons between the NNT for vaccination and the NNT for PHN treatment are methodologically problematic: they involve different outcome definitions, different patient populations, different time horizons, and different levels of evidence quality. The NNT for PHN treatment agents (approximately 4 to 8 for 50 percent pain reduction) describes analgesic benefit in patients who already have PHN, not prevention of its occurrence. Moreover, many PHN treatment trials share the same limitations of industry sponsorship and imperfect blinding as the vaccination trials. What can be said with reasonable confidence is that current pharmacological options for PHN are of limited and inconsistent efficacy, that many patients derive insufficient benefit, and that this treatment gap is clinically real. Whether that gap substantially changes the individual benefit-harm calculus of vaccination depends on personal risk tolerance, age, health status, and the values applied to transient vaccine-related discomfort versus the uncertain probability of developing refractory PHN.
Discussion
The ZOE trials produced relative efficacy estimates for RZV substantially higher than those observed for the prior live-attenuated vaccine across all age groups, with relative risk reductions of 91 to 97 percent. A central finding of this review, however, is that the near-uniform relative efficacy across age groups conceals meaningful variation in absolute benefit. The age-stratified ARR and NNT analysis presented here makes that variation visible and is directly relevant to individual clinical decision-making.
Among age groups, the 60 to 69 cohort shows the lowest trial-derived NNT (approximately 30) combined with the same NNH for grade 3 systemic reactions as the 50 to 59 group (approximately 11), reflecting the highest baseline shingles incidence in this age range. Adults aged 70 and older have a similar NNT (approximately 32) but a substantially higher NNH for severe reactions (approximately 25), reflecting the attenuated immune response to the adjuvant with advancing age. Adults aged 50 to 59 have the highest NNT (approximately 40) and the same NNH as the 60 to 69 group, reflecting lower baseline disease incidence despite similar relative efficacy. Whether any of these ratios represents a favorable, acceptable, or unfavorable benefit-harm profile is a value judgment that depends on individual circumstances and preferences, not a conclusion that can be drawn solely from the numbers. All three cohorts have NNTs substantially above 1, meaning most vaccinated individuals will not directly benefit from prevented shingles during the trial window, while a meaningful minority will experience grade 3 reactions.
The comparison of NNT and NNH requires careful framing that resists drawing premature conclusions. The adverse events captured by the NNH (grade 3 systemic reactions) are temporary, resolving within 1 to 3 days, and were not associated with lasting harm in the trial data. The benefit captured by the NNT is prevention of a condition that can range from mild to severely debilitating depending on individual factors including age, rash extent, and acute pain severity. As discussed above, PHN treatment is of limited and variable efficacy, and this is relevant context for individual decision-making. However, a reader should be aware that the NNT/NNH ratio itself does not weigh the severity or duration of harms against benefits, and that any QALY-based analysis rests on assumptions embedded in models funded by the manufacturer. ACIP cost-effectiveness analyses concluded that RZV meets acceptability thresholds, though such analyses are also sensitive to modeling assumptions.5
The conflict-of-interest and blinding concerns described in this review are not unique to RZV and reflect systemic features of the pharmaceutical regulatory landscape. The standard safeguards against sponsor influence include independent endpoint adjudication, pre-registered protocols, data and safety monitoring boards, and disclosure requirements. The ZOE program nominally met these standards for the primary efficacy endpoints.
However, the absence of independent funding, the structural impossibility of fully blinding a highly reactogenic adjuvanted vaccine against a clear placebo, the pervasive involvement of GSK employees in data handling and writing, and the open-label design of the long-term extension collectively mean that the evidence base for RZV rests on a narrower epistemic foundation than the headline relative efficacy figures suggest. Readers should weigh these limitations explicitly when drawing clinical conclusions, and should rely on absolute rather than relative effect measures when communicating benefit to individual patients.
Conclusions
Within the bounds of exclusively manufacturer-funded evidence, RZV demonstrated high relative efficacy against herpes zoster across all studied age groups over a 3.5-year trial horizon, with attenuation in the open-label long-term extension. Age-stratified NNT data show the lowest short-term absolute benefit in adults aged 50 to 59 and the most favorable balance of NNT versus NNH in adults aged 70 and older, where severe reactions are less frequent. Serious adverse events were not increased relative to placebo in the pivotal trials; the risk of Guillain-Barre syndrome identified post-marketing is rare.
The trials were observer-blind rather than double-blind, and the highly reactogenic adjuvant system makes participant-level inference of treatment assignment probable, potentially biasing solicited adverse event rates upward. All solicited safety data should be interpreted with this uncertainty in mind.
The pharmacological treatment options for established PHN are of incomplete and variable efficacy, and this informs but does not simplify the individual benefit-harm calculation. Clinicians should present both NNT and NNH data transparently, using age-appropriate estimates, and should acknowledge the structural limitations of the underlying evidence when discussing vaccination with patients.
The evidentiary limitations documented in this review do not permit a confident conclusion that the vaccine is either clearly net-beneficial or not, but they do identify the specific domains where additional independent research would most reduce uncertainty.
References
1. Cohen JI. Clinical practice: Herpes zoster. N Engl J Med. 2013;369(3):255-263. doi:10.1056/NEJMcp1302434
2. Harpaz R, Ortega-Sanchez IR, Seward JF; Advisory Committee on Immunization Practices Centers for Disease Control and Prevention. Prevention of herpes zoster: recommendations of the Advisory Committee on Immunization Practices. MMWR Recomm Rep. 2008;57(RR-5):1-30.
3. Johnson RW, Rice ASC. Clinical practice: Postherpetic neuralgia. N Engl J Med. 2014;371(16):1526-1533. doi:10.1056/NEJMcp1403062
4. Oxman MN, Levin MJ, Johnson GR, et al.; Shingles Prevention Study Group. A vaccine to prevent herpes zoster and postherpetic neuralgia in older adults. N Engl J Med. 2005;352(22):2271-2284. doi:10.1056/NEJMoa051016
5. Dooling KL, Guo A, Patel M, et al. Recommendations of the Advisory Committee on Immunization Practices for use of herpes zoster vaccines. MMWR Morb Mortal Wkly Rep. 2018;67(3):103-108. doi:10.15585/mmwr.mm6703a5
6. Lal H, Cunningham AL, Godeaux O, et al.; ZOE-50 Study Group. Efficacy of an adjuvanted herpes zoster subunit vaccine in older adults. N Engl J Med. 2015;372(22):2087-2096. doi:10.1056/NEJMoa1501184
7. Cunningham AL, Lal H, Kovac M, et al.; ZOE-70 Study Group. Efficacy of the herpes zoster subunit vaccine in adults 70 years of age or older. N Engl J Med. 2016;375(11):1019-1032. doi:10.1056/NEJMoa1603800
8. Boutry C, Hastie A, Diez-Domingo J, et al. The adjuvanted recombinant zoster vaccine confers long-term protection against herpes zoster: interim results of an extension study of the pivotal phase 3 clinical trials ZOE-50 and ZOE-70. Clin Infect Dis. 2022;74(8):1459-1467. doi:10.1093/cid/ciab629
9. Strezova A, Vongpunsawad S, Curran D, et al. Final analysis of the ZOE-LTFU trial to 11 years post-vaccination: efficacy of the adjuvanted recombinant zoster vaccine against herpes zoster and related complications. EClinicalMedicine. 2025;S2589-5370(25)00173-7. doi:10.1016/j.eclinm.2025.003
10. Therapeutics Initiative. Shingrix: a new vaccine for shingles. Therapeutics Letter. 2018;(114). Published October 2018. Accessed via NCBI Bookshelf: https://www.ncbi.nlm.nih.gov/books/NBK598509/
11. Mwakingwe-Omari A, Lecrenier N, Naficy A, et al. Recombinant zoster vaccine in immunocompetent and immunocompromised adults: a review of clinical studies. Hum Vaccin Immunother. 2023;19(3):2278362. doi:10.1080/21645515.2023.2278362
12. Izurieta HS, Wu X, Forshee R, et al. Recombinant zoster vaccine (Shingrix): real-world effectiveness in the first 2 years post-licensure. Clin Infect Dis. 2021;73(6):941-948. doi:10.1093/cid/ciab125
13. Fiore J, Co-van der Mee MM, Maldonado A, Glasser L, Watson P. Safety and reactogenicity of the adjuvanted recombinant zoster vaccine: experience from clinical trials and post-marketing surveillance. Ther Adv Vaccines Immunother. 2021;9:25151355211057479. doi:10.1177/25151355211057479
14. GlaxoSmithKline Biologicals. SHINGRIX (Zoster Vaccine Recombinant, Adjuvanted) [prescribing information]. Research Triangle Park, NC: GlaxoSmithKline; 2021.
15. Schulz KF, Altman DG, Moher D; CONSORT Group. CONSORT 2010 Statement: updated guidelines for reporting parallel group randomised trials. BMJ. 2010;340:c332. doi:10.1136/bmj.c332
16. Hrobjartsson A, Emanuelsson F, Skou Thomsen AS, Hilden J, Brorson S. Bias due to lack of patient blinding in clinical trials: a systematic review of trials randomizing patients to blind and nonblind sub-studies. Int J Epidemiol. 2014;43(4):1272-1283. doi:10.1093/ije/dyu115
17. Dworkin RH, Johnson RW, Breuer J, et al. Recommendations for the management of herpes zoster. Clin Infect Dis. 2007;44(Suppl 1):S1-26. doi:10.1086/510206
18. Gnann JW Jr, Whitley RJ. Herpes zoster. N Engl J Med. 2002;347(5):340-346. doi:10.1056/NEJMcp013211
19. Kim YJ, Lee CN, Agrawal SK, et al. Current scenario and future applicability of antivirals against herpes zoster. Viruses. 2022;14(12):2824. doi:10.3390/v14122824
20. Sacks GM. Unmet need in the treatment of postherpetic neuralgia. Am J Manag Care. 2013;19(1 Suppl):S207-213.
21. Fashner J, Bell AL. Herpes zoster and postherpetic neuralgia: prevention and management. Am Fam Physician. 2011;83(12):1432-1437.
22. Dworkin RH, Corbin AE, Young JP Jr, et al. Pregabalin for the treatment of postherpetic neuralgia: a randomized, placebo-controlled trial. Neurology. 2003;60(8):1274-1283. doi:10.1212/01.WNL.0000055433.55136.55
23. Nalamachu S, Morley-Forster P. Diagnosing and managing postherpetic neuralgia. Drugs Aging. 2012;29(11):863-869. doi:10.1007/s40266-012-0014-3